Sermorelin vs Ipamorelin: GH Secretagogue Mechanisms in Research

By Dr. Leonard Haberman, Chief Science Officer, OPTMZ Peptides Published: March 2, 2026 | Last Updated: April 15, 2026 What Are Sermorelin and Ipamorelin? Sermorelin and ipamorelin are growth hormone (GH) secretagogues — compounds that stimulate GH release from anterior pituitary somatotroph cells through distinct receptor-mediated mechanisms. In peptide research, they serve as representative model compounds for two separate GH regulatory pathways: the GHRH receptor pathway (sermorelin) and the ghrelin receptor pathway (ipamorelin). Researchers studying GH axis regulation select between these compounds based on which receptor mechanism is under investigation — they are not functionally interchangeable. Both compounds are available from OPTMZ Peptides as batch-verified research reagents, independently tested by Krause Analytical — DEA-registered, ISO/IEC 17025-certified. Batch-level Certificates of Analysis are published at the COA Vault → Understanding the GH Axis: GH, GHRH, and GHRP Growth hormone regulation involves three primary elements that frequently appear together in secretagogue research. Growth Hormone (GH / Somatotropin) is a 191-amino acid peptide hormone secreted by anterior pituitary somatotrophs. In pre-clinical research, GH is studied for its downstream effects on IGF-1 axis activation, lipid metabolism signaling, and cellular anabolic pathways. It is the effector molecule — the end product of the regulatory cascade that GHRH and GHRPs each modulate. Growth Hormone Releasing Hormone (GHRH) is a 44-amino acid neuropeptide produced in the arcuate nucleus of the hypothalamus. GHRH acts on the GHRH receptor (GHRHR) expressed on somatotroph cells, activating Gαs-coupled adenylyl cyclase, increasing intracellular cAMP, and driving GH synthesis and pulsatile secretion. Sermorelin is a synthetic GHRH analog that targets this pathway. Growth Hormone Releasing Peptides (GHRPs) are a structurally distinct class of synthetic secretagogues that stimulate GH release through a separate receptor — the GH secretagogue receptor type 1a (GHS-R1a), also known as the ghrelin receptor. GHRPs were first characterized in binding studies by Bowers CY in the 1980s–1990s and represent an independent intervention point for GH axis research (Bowers CY, Cell Mol Life Sci, 1998). Ipamorelin is the most receptor-selective GHRP studied to date. Research has demonstrated that simultaneous stimulation of both the GHRHR and GHS-R1a pathways produces synergistic GH release exceeding the additive effect of either compound alone — a finding that has made dual-pathway co-administration a standard experimental model in neuroendocrinology research (Sigalos JT, Pastuszak AW, Sex Med Rev, 2018). Sermorelin: GHRH Receptor Agonist in Research Molecular Structure and Receptor Mechanism Sermorelin is the N-terminal 29-amino acid fragment of native human GHRH — designated hGHRH[1-29]NH₂. This truncated fragment retains full biological activity at the GHRHR. The C-terminal residues beyond position 29 contribute to plasma stability but are not required for receptor binding or activation. The receptor mechanism proceeds as follows: sermorelin binds the extracellular domain of GHRHR on anterior pituitary somatotrophs, activating the Gαs protein-coupled adenylyl cyclase system. This increases intracellular cAMP, activating protein kinase A (PKA), which drives both acute GH exocytosis and GH gene transcription. The result is GH secretion that preserves the pulsatile pattern characteristic of physiologic GHRH-driven release (Walker RF, Clin Interv Aging, 2006). What Research Has Examined Sermorelin has been investigated as a model compound for GHRH axis studies in aging research models, where age-associated decline in hypothalamic GHRH output has been documented in rodent studies. Pre-clinical research has also used sermorelin to probe somatotroph cell population integrity and GHRHR responsiveness under various experimental conditions. A practical advantage of sermorelin over synthetic GHRH(1-44) as a research tool is its metabolic stability. The 29-residue fragment demonstrates extended plasma half-life in animal models compared to the native 44-residue peptide, making it a more tractable compound for in vivo GHRH receptor studies. OPTMZ Peptides supplies Sermorelin as a batch-verified research compound. Current batch purity data is available at the COA Vault → Ipamorelin: GHS-R1a Agonist and Selective GHRP Molecular Structure and Receptor Mechanism Ipamorelin is a pentapeptide with the sequence Aib-His-D-2-Nal-D-Phe-Lys-NH₂, first fully characterized by Raun et al. in 1998 as a selective GH secretagogue (Raun K et al., Eur J Endocrinol, 1998). It acts as a GHS-R1a agonist, mimicking the GH-releasing component of ghrelin’s activity without the broader metabolic receptor engagement associated with endogenous ghrelin. Unlike earlier GHRPs — GHRP-2 and GHRP-6 — ipamorelin activates the GHS-R1a without significant co-stimulation of ACTH, cortisol, or prolactin secretion in pre-clinical models at GH-stimulating concentrations. This selectivity profile makes ipamorelin the preferred GHS-R1a tool compound in research designs where HPA axis crosstalk would introduce confounding variables. Ipamorelin Selectivity Compared to Earlier GHRPs The original characterization study (Raun et al., 1998) demonstrated that ipamorelin produced GH release in rats comparable to GHRP-6 on a molar basis, while exhibiting significantly lower stimulation of ACTH and cortisol. This was attributed to ipamorelin’s cleaner binding profile at GHS-R1a versus the less selective receptor interactions of GHRP-2 and GHRP-6. For research requiring isolated GHS-R1a → GH axis activation, this selectivity is a critical methodological consideration. Ipamorelin in CJC-1295 Combination Models In pre-clinical research, ipamorelin is frequently studied in combination with GHRH analogs — most commonly CJC-1295 (a DAC-modified GHRH analog with extended half-life) — to examine the synergistic effects of simultaneous GHRHR and GHS-R1a activation on GH pulse amplitude and frequency. This dual-pathway experimental design has become a standard model for studying GH secretion dynamics. OPTMZ Peptides supplies both Ipamorelin and CJC-1295 + Ipamorelin as batch-verified research compounds, independently tested by Krause Analytical. Batch COAs are available at the COA Vault → Sermorelin vs Ipamorelin: Mechanistic Comparison Property Sermorelin Ipamorelin Compound class GHRH analog GHRP (ghrelin mimetic) Primary receptor GHRHR (Gαs-coupled) GHS-R1a (Gαq/11-coupled) Signaling cascade cAMP → PKA IP₃ / DAG → Ca²⁺ GH release pattern Pulsatile (physiologic) Pulsatile (acute burst) Selectivity vs HPA axis Neutral High (low ACTH/cortisol co-stimulation) Peptide length 29 amino acids 5 amino acids (pentapeptide) Primary research application GHRH axis regulation studies Selective GHS-R1a agonism models Combination research use Paired with GHRPs (ipamorelin, GHRP-6) Paired with GHRH analogs (CJC-1295) These mechanistic differences define distinct research applications. Sermorelin is the appropriate tool compound when the research question concerns GHRH receptor integrity, GHRH-mediated cAMP
TB-500 and Hair Growth Research: What Current Studies Suggest About Follicular Mechanisms
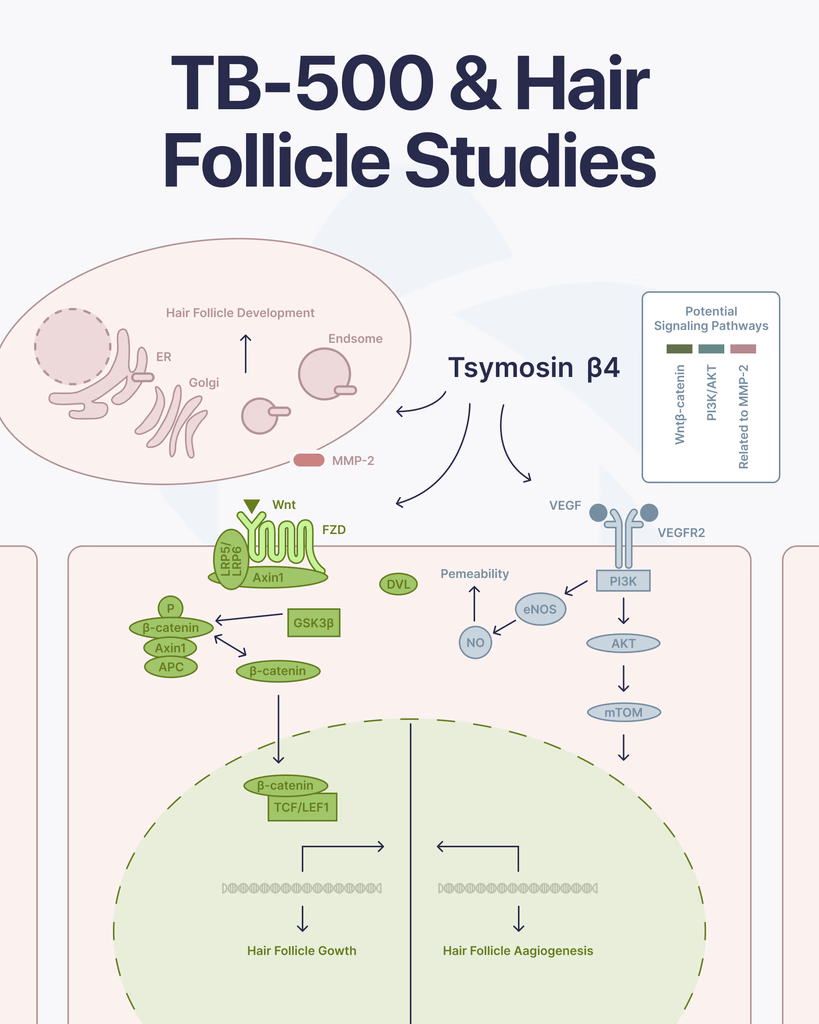
By Dr. Leonard Haberman, Chief Science Officer, OPTMZ Peptides Published: March 2,2026 | Last Updated: April 14, 2026 TB-500 is a synthetic analogue of Thymosin Beta-4 (Tβ4), a 43-amino acid peptide naturally expressed in mammalian tissue. Pre-clinical and in vitro research has examined its role in cell migration, angiogenesis, and tissue repair. More recently, researchers have investigated whether Tβ4-related mechanisms extend to hair follicle biology — an area supported by several published studies, primarily conducted in animal models and isolated cell cultures. What Is TB-500? Molecular Background for Research Contexts TB-500 is derived from the C-terminal fragment of Thymosin Beta-4. Its molecular structure centers on a conserved actin-binding domain (LKKTET sequence), which is understood to regulate actin dynamics — specifically, the polymerization of G-actin into F-actin filaments that govern cellular migration and morphology. In research settings, Tβ4 has been studied across several biological systems, including wound healing models, cardiac tissue, and — relevant to this review — hair follicle regeneration pathways. TB-500’s designation as a synthetic research peptide reflects its use as an accessible laboratory tool for investigating the activity of native Tβ4 in controlled pre-clinical experimental contexts. OPTMZ Peptides supplies TB-500 as a lyophilized research peptide, independently tested by Krause Analytical (DEA-registered, ISO/IEC 17025-certified, Austin TX). Batch purity is verified by HPLC to a minimum of 98%, with most batches returning 98.5–99.9%. Current batch COA data is available in the OPTMZ COA Vault. Hair Follicle Biology: Why Researchers Are Examining Tβ4 Activity Hair follicle cycling involves three principal phases: anagen (active growth), catagen (regression), and telogen (rest). Transition from telogen back into anagen requires coordinated signaling among follicular stem cells, dermal papilla cells, and supporting vascular structures. Disruption at any stage — through reduced stem cell activation, diminished vascularization, or inflammatory signaling — is associated with arrested cycling in research models. Several of the biological pathways Thymosin Beta-4 has been studied for — stem cell migration, vascular endothelial growth factor (VEGF) signaling, and regulation of actin dynamics — overlap with the cellular events that govern this follicular cycle. This mechanistic overlap is the primary reason researchers have examined whether Tβ4 influences hair follicle behavior in experimental contexts. What Studies Have Examined: TB-500 and Hair Follicle Mechanisms Thymosin Beta-4 and Hair Follicle Stem Cell Activation The foundational study in this area was published by Philp et al. (2004) in the FASEB Journal. Researchers investigated the effect of exogenous Thymosin Beta-4 on hair follicle cycling in murine models. The study found that Tβ4 administration was associated with accelerated transition into the anagen phase and attributed this effect, in part, to activation of hair follicle stem cells. The authors concluded that Tβ4 influences critical events in the active phase of the hair follicle cycle, including stem cell mobilization and cell migration toward the dermal papilla (Philp et al., 2004 — PMID 14657002). This study has been cited 166 times and remains the primary reference point for subsequent investigations into Tβ4 and follicular biology. Angiogenesis, VEGF Signaling, and Vascular Support of Follicles Adequate vascularization of the hair follicle bulb is required for nutrient delivery during the anagen phase. Research has examined Thymosin Beta-4’s role in promoting angiogenesis — the formation of new blood vessels from existing vasculature — as a mechanism with potential relevance to follicular support. Philp et al. (2004) in Mechanisms of Development documented that Tβ4 promotes angiogenesis and noted its observed association with hair follicle growth in murine models, connecting the peptide’s vascular activity to follicular biology (Philp et al., 2004 — PMID 15210177). Tβ4’s upregulation of VEGF expression — documented in separate vascular research contexts — is understood to be the mechanistic pathway underlying this angiogenic effect in pre-clinical models. Mechanism Investigation: The 2015 PLOS One Study A 2015 study by Gao et al. published in PLOS One set out specifically to investigate the mechanism by which Thymosin Beta-4 induces hair growth in mouse models. Prior studies had established the association; this study examined the downstream molecular events. The investigators found evidence that Tβ4 influences both stem cell migration and follicular development through pathways involving the Wnt signaling cascade, a known regulator of hair follicle morphogenesis and cycling (Gao et al., 2015 — PMID 26076359). Tβ4 Overexpression and Follicular Morphogenesis Cha et al. (2010) examined what occurs when Thymosin Beta-4 is overexpressed rather than supplemented in controlled models, finding that excess Tβ4 was associated with abnormal hair and tooth morphogenesis — suggesting the peptide operates within regulated concentration ranges relevant to normal follicular architecture (Cha et al., 2010 — PMID 20013654). This finding is methodologically important for researchers designing Tβ4 experimental protocols. Secondary Follicle Research Beyond the primary murine and in vitro models, Dai et al. (2021) examined the role of Thymosin Beta-4 in the biology of secondary hair follicles in goat cashmere production — a distinct but mechanistically relevant model for understanding Tβ4’s influence on follicle initiation and fiber growth pathways (Dai et al., 2021 — PMC7875905). What Does This Research Establish — and What Does It Not? The published pre-clinical literature establishes a coherent mechanistic framework: Thymosin Beta-4 influences hair follicle biology through stem cell activation, angiogenesis, and Wnt pathway modulation in animal models and isolated cell systems. The primary studies are well-cited, methodologically documented, and consistently cited in peer-reviewed reviews of follicular biology. What this research does not establish: Human clinical evidence is limited. The majority of studies have been conducted in murine models or isolated cells. Controlled human trials examining Tβ4 and hair follicle outcomes have not been published at the level of randomized controlled evidence. TB-500 as a synthetic analogue has been studied primarily as a research tool for investigating native Tβ4 activity in pre-clinical settings. Research-grade TB-500 is not a pharmaceutical product, is not FDA-approved, and is supplied exclusively for laboratory use. Dose-response relationships in follicular research remain incompletely characterized. The Cha et al. overexpression data indicates that concentration level is a relevant variable in experimental design. Researchers accessing TB-500 for follicular or related studies should treat the current
Peptide Mass Spectrometry: How Identity Verification Works in Research-Grade Compound Supply

By Dr. Leonard Haberman, Chief Science Officer, OPTMZ Peptides Published: March 2,2026 | Last Updated: April 13, 2026 Peptide mass spectrometry is an analytical technique that determines the molecular identity of a peptide by measuring the mass-to-charge ratio of its ionized fragments. In research-grade compound supply, it is the definitive method for confirming that a peptide’s amino acid sequence matches its specified structure — a function that HPLC purity analysis alone cannot perform. What Is Peptide Mass Spectrometry? Mass spectrometry (MS) measures the mass-to-charge ratio (m/z) of ionized molecules. When applied to peptides, this produces a spectrum of mass values that functions as a molecular fingerprint: each peptide generates a unique pattern of fragment ions that corresponds to its specific amino acid sequence and molecular weight. In the context of research-grade peptide verification, mass spectrometry answers a question that purity data cannot: is this compound actually the peptide it is labeled as? A sample can achieve 99% purity by HPLC and still be the wrong compound — or a structurally similar analog — if identity confirmation is not independently performed. Mass spectrometry closes that verification gap. The technique was first systematically applied to peptide fragmentation analysis by Roepstorff and Fohlman, whose 1984 nomenclature for peptide fragment ion series (b-ions and y-ions) established the interpretive framework still in use today (Roepstorff P & Fohlman J, 1984, PMID: 6525415). How Does Mass Spectrometry Identify a Peptide? The identification process proceeds in three stages: 1. Ionization The peptide sample must be converted to gas-phase ions before mass measurement. The two dominant ionization methods for peptides — ESI and MALDI — are described in detail below. Ionization method selection affects sensitivity, throughput, and the types of structural information extractable from the spectrum. 2. Mass separation Ions are separated in a mass analyzer based on their m/z ratio. Common analyzer types in peptide MS include quadrupole, time-of-flight (TOF), and ion trap instruments. Each offers different trade-offs between mass accuracy, resolution, and scan speed. For high-resolution research-grade identity confirmation, TOF and Orbitrap analyzers are typically used due to their sub-ppm mass accuracy. 3. Detection and spectrum interpretation The detector produces a spectrum of ion abundances across m/z values. For intact peptide identity confirmation, the molecular ion peak is matched against the theoretical mass of the target compound. For sequence-level confirmation via tandem MS, the fragment ion series is mapped against predicted b-ion and y-ion series for the stated amino acid sequence. A match between observed and theoretical mass within the instrument’s accepted error tolerance (typically ≤5 ppm for high-resolution instruments) constitutes identity confirmation. Deviation beyond this tolerance indicates a structural mismatch, degradation product, or synthesis error — all of which constitute grounds for batch rejection in a quality-controlled supply chain. What Is the Difference Between ESI and MALDI for Peptide Analysis? The two primary ionization methods used in peptide mass spectrometry each have distinct operational characteristics and applications: Electrospray Ionization (ESI) ESI converts peptides in solution directly to gas-phase ions by passing the liquid sample through a charged capillary under atmospheric pressure. The process generates multiply charged ions, which is particularly useful for larger peptides — the multiple charge states allow heavier molecules to appear at lower m/z values within the instrument’s detection range. ESI is highly compatible with liquid chromatography (LC), making LC-MS and LC-MS/MS the dominant platform for comprehensive peptide analysis. The continuous-flow nature of ESI allows real-time coupling with separation systems, enabling simultaneous purity profiling and identity confirmation in a single analytical run. Yates, Ruse, and Nakorchevsky (2009) provide a comprehensive overview of LC-MS/MS workflows in proteomics and research compound characterization (PMID: 19838170). At Krause Analytical, ESI-based mass spectrometry is used as part of the identity confirmation step in OPTMZ Peptides’ testing panel — conducted on every batch submitted for quality verification, independently of purity analysis. Matrix-Assisted Laser Desorption/Ionization (MALDI) MALDI embeds the peptide sample in a crystalline matrix material that absorbs laser energy. When the laser fires, the matrix transfers that energy to the peptide molecules, desorbing and ionizing them from the solid surface. MALDI typically produces singly charged ions and is particularly well-suited to rapid molecular weight determination and peptide mass fingerprinting (PMF). MALDI instruments — especially MALDI-TOF platforms — are widely used in high-throughput screening environments due to their speed and relative tolerance for sample contaminants. However, for sequence-level confirmation requiring tandem fragmentation, ESI-based systems are generally preferred in research-grade supply chain applications due to their direct compatibility with MS/MS workflows. What Does MS/MS (Tandem Mass Spectrometry) Add to Peptide Sequencing? Standard MS provides a molecular weight measurement — confirmation that the peptide falls within the expected mass range. Tandem mass spectrometry (MS/MS) goes further: it fragments the parent ion and analyzes the resulting fragment ions to reconstruct the amino acid sequence directly. The MS/MS process works as follows: Precursor ion selection: A specific m/z value (the molecular ion of interest) is isolated in the first mass analyzer stage Fragmentation: The isolated ion is subjected to collision-induced dissociation (CID), electron transfer dissociation (ETD), or higher-energy collisional dissociation (HCD), breaking the peptide backbone at specific sites Fragment ion analysis: The resulting fragment ions are analyzed in the second stage, generating a spectrum of b-ions (N-terminal fragments) and y-ions (C-terminal fragments) Sequence reconstruction: The mass differences between adjacent b-ions or y-ions correspond to the masses of individual amino acid residues, allowing direct sequence readout Syka et al. (2004) demonstrated the utility of electron transfer dissociation for sequence analysis of peptides and proteins, establishing an important methodological extension to conventional CID-based MS/MS (PMID: 15258601). For research-grade peptide suppliers, MS/MS is the gold standard for verifying that a compound’s primary structure — its actual amino acid sequence — matches the label claim, not merely its molecular weight. How Is Mass Spectrometry Used in Research-Grade Peptide Quality Control? In a controlled peptide supply chain, mass spectrometry performs two non-redundant functions that together constitute complete identity verification: Function 1: Intact mass confirmation The intact molecular ion mass is compared to the