By Dr. Leonard Haberman, Chief Science Officer, OPTMZ Peptides Published: March 2,2026 | Last Updated: April 14, 2026
TB-500 is a synthetic analogue of Thymosin Beta-4 (Tβ4), a 43-amino acid peptide naturally expressed in mammalian tissue. Pre-clinical and in vitro research has examined its role in cell migration, angiogenesis, and tissue repair. More recently, researchers have investigated whether Tβ4-related mechanisms extend to hair follicle biology — an area supported by several published studies, primarily conducted in animal models and isolated cell cultures.
What Is TB-500? Molecular Background for Research Contexts
TB-500 is derived from the C-terminal fragment of Thymosin Beta-4. Its molecular structure centers on a conserved actin-binding domain (LKKTET sequence), which is understood to regulate actin dynamics — specifically, the polymerization of G-actin into F-actin filaments that govern cellular migration and morphology.
In research settings, Tβ4 has been studied across several biological systems, including wound healing models, cardiac tissue, and — relevant to this review — hair follicle regeneration pathways. TB-500’s designation as a synthetic research peptide reflects its use as an accessible laboratory tool for investigating the activity of native Tβ4 in controlled pre-clinical experimental contexts.
OPTMZ Peptides supplies TB-500 as a lyophilized research peptide, independently tested by Krause Analytical (DEA-registered, ISO/IEC 17025-certified, Austin TX). Batch purity is verified by HPLC to a minimum of 98%, with most batches returning 98.5–99.9%. Current batch COA data is available in the OPTMZ COA Vault.
Hair Follicle Biology: Why Researchers Are Examining Tβ4 Activity
Hair follicle cycling involves three principal phases: anagen (active growth), catagen (regression), and telogen (rest). Transition from telogen back into anagen requires coordinated signaling among follicular stem cells, dermal papilla cells, and supporting vascular structures. Disruption at any stage — through reduced stem cell activation, diminished vascularization, or inflammatory signaling — is associated with arrested cycling in research models.
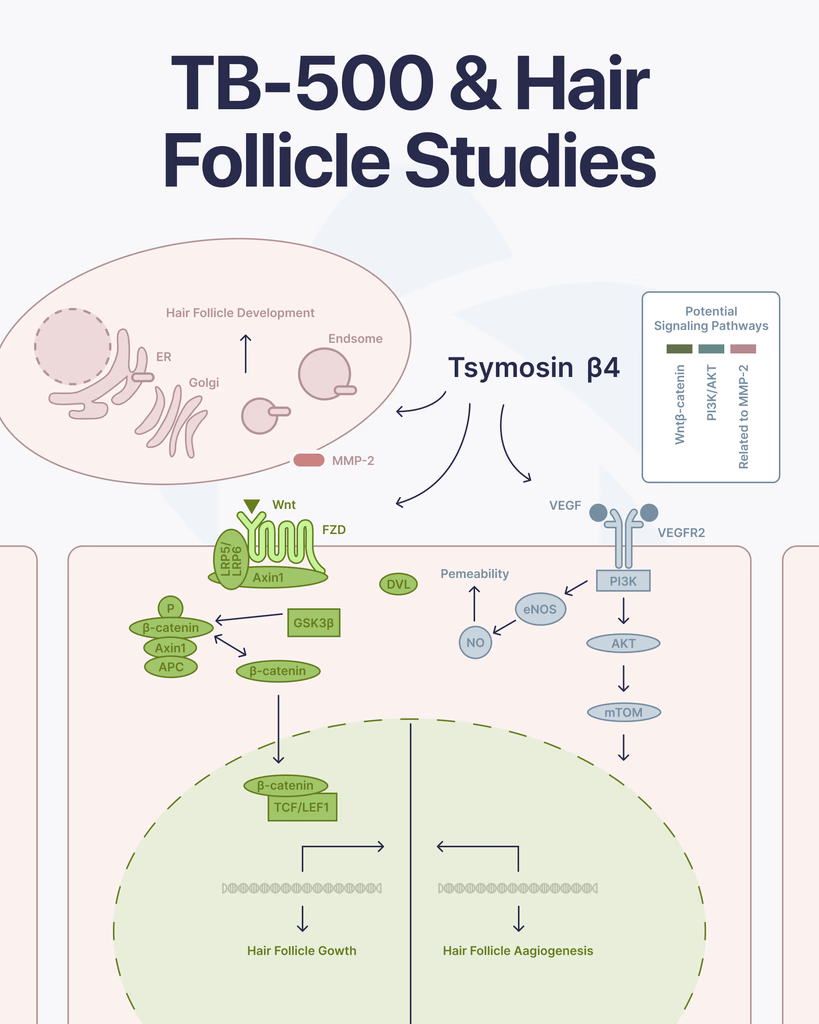
Several of the biological pathways Thymosin Beta-4 has been studied for — stem cell migration, vascular endothelial growth factor (VEGF) signaling, and regulation of actin dynamics — overlap with the cellular events that govern this follicular cycle. This mechanistic overlap is the primary reason researchers have examined whether Tβ4 influences hair follicle behavior in experimental contexts.
What Studies Have Examined: TB-500 and Hair Follicle Mechanisms
Thymosin Beta-4 and Hair Follicle Stem Cell Activation
The foundational study in this area was published by Philp et al. (2004) in the FASEB Journal. Researchers investigated the effect of exogenous Thymosin Beta-4 on hair follicle cycling in murine models. The study found that Tβ4 administration was associated with accelerated transition into the anagen phase and attributed this effect, in part, to activation of hair follicle stem cells. The authors concluded that Tβ4 influences critical events in the active phase of the hair follicle cycle, including stem cell mobilization and cell migration toward the dermal papilla (Philp et al., 2004 — PMID 14657002).
This study has been cited 166 times and remains the primary reference point for subsequent investigations into Tβ4 and follicular biology.
Angiogenesis, VEGF Signaling, and Vascular Support of Follicles
Adequate vascularization of the hair follicle bulb is required for nutrient delivery during the anagen phase. Research has examined Thymosin Beta-4’s role in promoting angiogenesis — the formation of new blood vessels from existing vasculature — as a mechanism with potential relevance to follicular support.
Philp et al. (2004) in Mechanisms of Development documented that Tβ4 promotes angiogenesis and noted its observed association with hair follicle growth in murine models, connecting the peptide’s vascular activity to follicular biology (Philp et al., 2004 — PMID 15210177). Tβ4’s upregulation of VEGF expression — documented in separate vascular research contexts — is understood to be the mechanistic pathway underlying this angiogenic effect in pre-clinical models.
Mechanism Investigation: The 2015 PLOS One Study
A 2015 study by Gao et al. published in PLOS One set out specifically to investigate the mechanism by which Thymosin Beta-4 induces hair growth in mouse models. Prior studies had established the association; this study examined the downstream molecular events. The investigators found evidence that Tβ4 influences both stem cell migration and follicular development through pathways involving the Wnt signaling cascade, a known regulator of hair follicle morphogenesis and cycling (Gao et al., 2015 — PMID 26076359).
Tβ4 Overexpression and Follicular Morphogenesis
Cha et al. (2010) examined what occurs when Thymosin Beta-4 is overexpressed rather than supplemented in controlled models, finding that excess Tβ4 was associated with abnormal hair and tooth morphogenesis — suggesting the peptide operates within regulated concentration ranges relevant to normal follicular architecture (Cha et al., 2010 — PMID 20013654). This finding is methodologically important for researchers designing Tβ4 experimental protocols.
Secondary Follicle Research
Beyond the primary murine and in vitro models, Dai et al. (2021) examined the role of Thymosin Beta-4 in the biology of secondary hair follicles in goat cashmere production — a distinct but mechanistically relevant model for understanding Tβ4’s influence on follicle initiation and fiber growth pathways (Dai et al., 2021 — PMC7875905).
What Does This Research Establish — and What Does It Not?
The published pre-clinical literature establishes a coherent mechanistic framework: Thymosin Beta-4 influences hair follicle biology through stem cell activation, angiogenesis, and Wnt pathway modulation in animal models and isolated cell systems. The primary studies are well-cited, methodologically documented, and consistently cited in peer-reviewed reviews of follicular biology.
What this research does not establish:
- Human clinical evidence is limited. The majority of studies have been conducted in murine models or isolated cells. Controlled human trials examining Tβ4 and hair follicle outcomes have not been published at the level of randomized controlled evidence.
- TB-500 as a synthetic analogue has been studied primarily as a research tool for investigating native Tβ4 activity in pre-clinical settings. Research-grade TB-500 is not a pharmaceutical product, is not FDA-approved, and is supplied exclusively for laboratory use.
- Dose-response relationships in follicular research remain incompletely characterized. The Cha et al. overexpression data indicates that concentration level is a relevant variable in experimental design.
Researchers accessing TB-500 for follicular or related studies should treat the current literature as preliminary pre-clinical evidence — informative for hypothesis formation and experimental design, not as established clinical outcomes.
TB-500 Research Peptide: Batch Verification Data
OPTMZ Peptides sources TB-500 exclusively from verified synthesis batches that undergo a seven-method analytical testing panel at Krause Analytical (DEA-registered, ISO/IEC 17025 accredited, Austin TX) before entering inventory. Methods include HPLC purity analysis, mass spectrometry identity confirmation, endotoxin (LAL), heavy metals (ICP-MS), microbial screening, pH stability, and visual inspection.
Current batch purity data for TB-500 is published and searchable in the OPTMZ COA Vault. Researchers can cross-reference batch numbers from vial labels against published COA records. For full methodology documentation, see How OPTMZ Tests Research Peptides.
TB-500 is available in multiple vial configurations. The TB-500 product page includes current batch specifications and Lab Analysis tab documentation.
This product is intended strictly for in-vitro laboratory research conducted by qualified professionals. It is not intended for human or animal consumption, clinical use, or therapeutic application. Research Use Only. Not evaluated by the FDA.
By Dr. Leonard Haberman, Chief Science Officer, OPTMZ Peptides. Dr. Haberman oversees analytical quality assurance at OPTMZ Peptides, with expertise in HPLC-based purity verification, mass spectrometry identity confirmation, and third-party laboratory validation for research-grade peptide compounds.