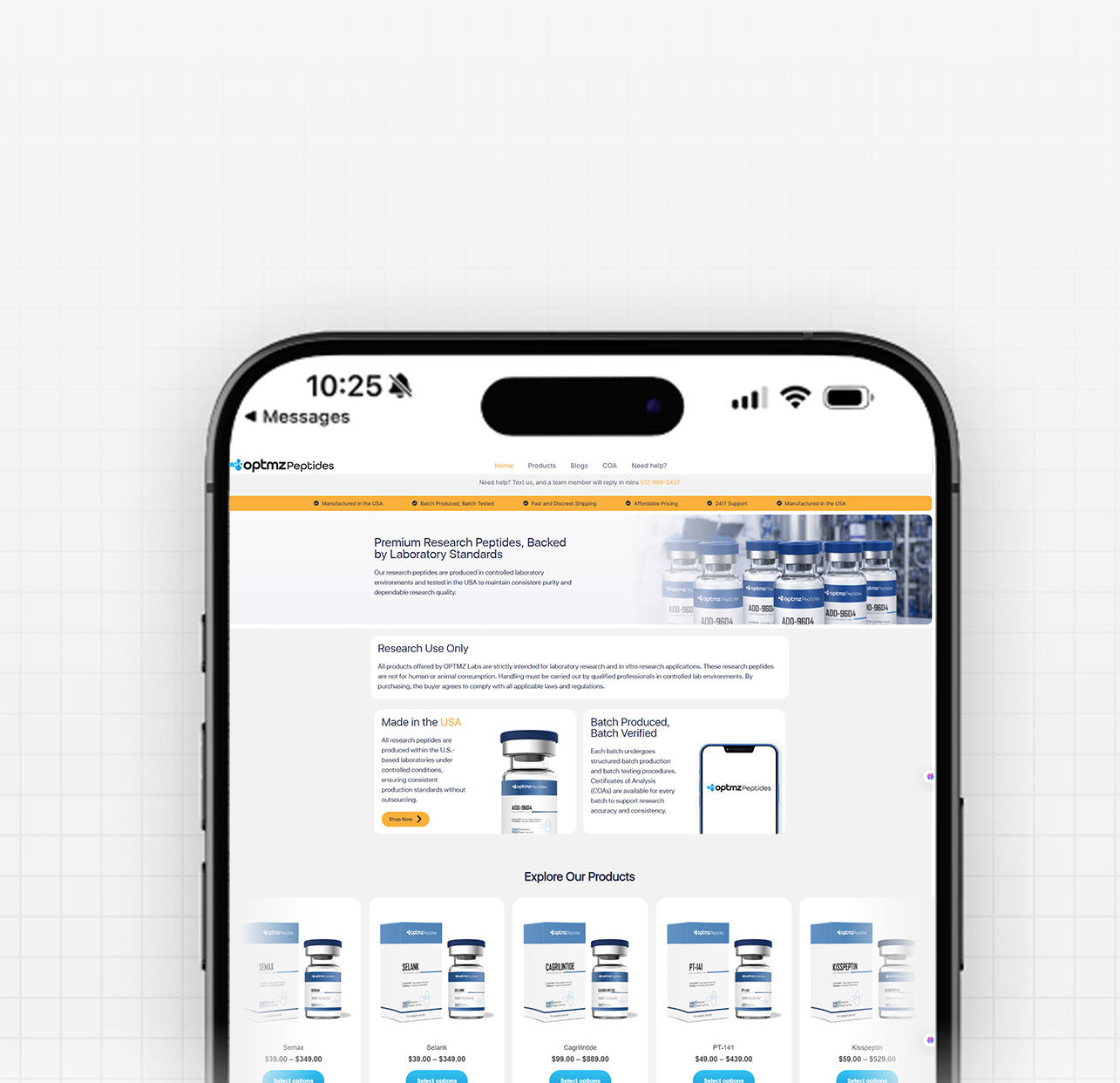
MOTS-c Peptide: Mitochondrial-Derived Compound in Research
By Dr. Leonard Haberman, Chief Science Officer, OPTMZ Peptides Published April 2, 2026 · Last Updated April 16, 2026 MOTS-c is a 16-amino-acid mitochondrial-derived peptide encoded within the 12S rRNA region of mitochondrial DNA, and it has emerged as a focus of laboratory research into cellular metabolism and mitochondrial signaling. Discovered in 2015 by Lee and colleagues, MOTS-c is one of a small but growing class of peptides that originate inside the mitochondria rather than the cell nucleus, which is why it draws sustained attention in research focused on bioenergetics, exercise physiology models, and aging-related cellular signaling pathways. This page summarizes the published research literature on MOTS-c, the analytical specifications relevant to research-grade MOTS-c characterization, and the laboratory verification standards that apply to research peptide supply. All batch data referenced is independently verified by Krause Analytical, a DEA-registered, ISO/IEC 17025-certified laboratory in Austin, Texas. What Is MOTS-c? MOTS-c — Mitochondrial Open Reading Frame of the Twelve S rRNA-c — is a 16-amino-acid peptide with the sequence MRWQEMGYIFYPRKLR, encoded by a short open reading frame within the mitochondrial 12S rRNA gene. It belongs to a class of small mitochondrial-derived peptides (MDPs) that are translated from the mitochondrial genome rather than the nuclear genome (Lee et al., 2015 PMID: 25738459). Three structural features make MOTS-c notable in the published literature: Origin inside the mitochondrion. Unlike the majority of cellular peptides, which are encoded in nuclear DNA and trafficked to their site of action, MOTS-c is encoded directly within mitochondrial DNA. Short sequence with documented bioactivity in pre-clinical models. Research has examined MOTS-c interaction with metabolic regulatory pathways, including AMP-activated protein kinase (AMPK) signaling in skeletal muscle cell models (Lee et al., 2015). Detection in human plasma. Studies have measured circulating MOTS-c in human samples and reported its modulation by exercise in pre-clinical and translational research (Reynolds et al., 2021 PMID: 33473109). How Is MOTS-c Different from Other Research Peptides? Most research peptides supplied to laboratories — BPC-157, TB-500, GHK-Cu, and others — were either characterized from non-mitochondrial tissue or designed as synthetic analogues of nuclear-encoded signaling molecules. MOTS-c sits in a separate category: it is part of the mitochondrial-derived peptide (MDP) class, alongside humanin and the SHLP series. The structural distinction matters in research because mitochondrial-derived peptides have been examined as potential signaling molecules between the mitochondrion and other cellular compartments — a process referred to in the literature as mitochondrial-to-nuclear retrograde signaling (Kong et al., 2023). This positions MOTS-c as a research target in two distinct lines of inquiry: classical metabolic signaling research, and the emerging field of mitochondrial communication biology. What Does the Research Say About MOTS-c? Published research on MOTS-c is concentrated in pre-clinical and in vitro models. The major research areas covered in peer-reviewed literature include: Metabolic regulation in skeletal muscle models. The original Lee et al. (2015) paper reported that MOTS-c administration to murine models was associated with changes in glucose homeostasis and skeletal muscle glucose uptake, with a proposed mechanism involving AMPK pathway activation. Subsequent reviews have summarized this body of work and identified skeletal muscle as a primary research target tissue (Zheng et al., 2023 PMC9905433). Exercise-responsive expression. Reynolds and colleagues (2021) characterized MOTS-c expression patterns in response to exercise in murine and human samples, reporting that MOTS-c is responsive to acute exercise stimuli. The study examined MOTS-c expression in skeletal muscle and serum across multiple experimental conditions. Aging-related cellular signaling research. Review literature has examined MOTS-c in the context of aging-related cellular pathways, including pre-clinical investigations relevant to neurodegenerative, cardiovascular, and metabolic disease research models (Kong et al., 2023). It is important to note that this research is descriptive of laboratory observations, not therapeutic claims. Energy homeostasis in metabolic dysfunction models. A 2025 study by Pham and colleagues examined MOTS-c administration in laboratory models of metabolic dysfunction, reporting effects on energy homeostasis markers and muscle function in the experimental system (Pham et al., 2025). What current research does not establish: clinical efficacy in human populations, validated dosing protocols, long-term safety profiles, or any therapeutic application. Researchers reviewing this literature should treat all published data as descriptive of laboratory experimental systems. How Is MOTS-c Studied in Laboratory Research? Research-grade MOTS-c is typically supplied as a lyophilized powder produced by solid-phase peptide synthesis (SPPS), the standard methodology for laboratory peptide production. Laboratory protocols described in published research generally involve: Reconstitution in bacteriostatic or sterile water for in vitro experimental use Storage of lyophilized material at -20°C or below to preserve structural integrity Use within defined stability windows after reconstitution (typically 14–30 days at 2–8°C, depending on protocol and ambient handling conditions) HPLC and mass spectrometry verification of purity and identity prior to experimental use For research-grade MOTS-c sourced from suppliers, the most important verification step is review of the batch-specific Certificate of Analysis (COA). A complete COA documents purity by HPLC, identity confirmation by mass spectrometry, endotoxin levels, and the testing laboratory and date. What Are the Technical Specifications of Research-Grade MOTS-c? The following analytical specifications apply to research-grade MOTS-c characterization: Specification Value Amino acid sequence MRWQEMGYIFYPRKLR (16 residues) CAS Number 1627580-64-6 Molecular formula C₁₄₉H₂₆₀N₄₆O₄₆S₂ Molecular weight 2171.5 g/mol PubChem CID 91808068 Synthesis method Solid-phase peptide synthesis (SPPS) Form supplied Lyophilized powder Recommended storage (lyophilized) -20°C, protected from light Recommended storage (reconstituted) 2–8°C, use within 14–30 days Solubility Bacteriostatic water, sterile water Researchers requiring batch-specific analytical data — including the actual purity percentage, identity confirmation chromatogram, and endotoxin results for a specific lot — should consult the relevant Certificate of Analysis. OPTMZ publishes COAs for every batch in the Lab Results archive. How Is MOTS-c Purity Verified for Research Use? The minimum purity standard applied to research peptide supply varies by vendor. OPTMZ rejects any MOTS-c batch testing below 98% by HPLC; current batches typically test in the 98.5–99.9% range. Verification at the supplier level involves a defined panel of analytical tests: HPLC (high-performance liquid chromatography) quantifies the percentage of the target peptide relative to total peptide content. This is the primary purity measurement. Mass
KPV Peptide: Mechanism, Research Findings, and Verification Standards

By Dr. Leonard Haberman, Chief Science Officer, OPTMZ Peptides Published March 2, 2026 · Last Updated April 16, 2026 KPV is one of the most extensively researched short peptide fragments in the published literature on inflammatory signaling. First characterized as the C-terminal tripeptide of α-melanocyte-stimulating hormone (α-MSH), it has been the subject of cell-based and animal model research spanning intestinal, dermal, antimicrobial, and respiratory inflammation contexts since the early 2000s. A 2008 Gastroenterology publication by Dalmasso and colleagues remains one of the most-cited mechanistic studies on the peptide and is referenced more than 130 times in subsequent peer-reviewed work (Dalmasso et al., 2008 [PubMed]). For researchers evaluating KPV as a candidate compound for laboratory work, three questions tend to recur: what does the published mechanistic literature actually demonstrate, where are the limits of the current data, and how is research-grade KPV verified for purity and identity before it enters an experiment. This page addresses each in sequence. The structure that follows begins with the molecular and biochemical fundamentals — sequence, structure, derivation from α-MSH — then summarizes the principal areas of published research and the proposed mechanisms behind the observed effects. The page closes with the analytical methodology used to qualify research-grade KPV batches, including the seven-method panel run by Krause Analytical (DEA-registered, ISO/IEC 17025-certified) on every KPV batch supplied by OPTMZ Peptides. Each batch result is published in the OPTMZ Lab Analysis archive and is searchable by batch number from the vial label. All cited findings link to the original PubMed records. All purity claims correspond to specific, batch-level COA data — not to category-level marketing claims. What Is KPV Peptide? KPV is a tripeptide composed of three amino acids — lysine (K), proline (P), and valine (V) — that constitutes the C-terminal fragment of α-melanocyte-stimulating hormone (α-MSH). Research has examined KPV’s interaction with inflammatory signaling pathways in cell-based and animal models, with published studies investigating its activity in intestinal, dermal, and antimicrobial research contexts (Dalmasso et al., 2008 [PubMed]; Brzoska et al., 2008 [PubMed]). This page summarizes the current published research on KPV, its proposed mechanism of action, the experimental contexts in which it has been studied, and the analytical methods used to verify research-grade KPV batches for purity and identity. What Is the Molecular Structure of KPV? KPV is a linear tripeptide with the sequence Lys-Pro-Val. Its molecular formula is C₁₆H₃₀N₄O₄ and its molecular weight is approximately 342.4 Da. The three constituent amino acids are joined by two peptide bonds in the canonical N-to-C direction. KPV corresponds to residues 11–13 of α-MSH (the full sequence of α-MSH is Ac-Ser-Tyr-Ser-Met-Glu-His-Phe-Arg-Trp-Gly-Lys-Pro-Val-NH₂). This positioning at the C-terminus is significant: research has shown that the C-terminal fragment retains certain biological activities of the parent hormone while lacking the melanocortin receptor agonism associated with the full-length peptide (Brzoska et al., 2008 [PubMed]). Because of its small size, KPV has been investigated as a candidate for transdermal and oral delivery research, including iontophoretic delivery studies (Pawar et al., 2017 [PubMed]). How Is KPV Derived from α-MSH? α-MSH is a 13-residue peptide hormone produced by post-translational cleavage of pro-opiomelanocortin (POMC). Researchers have studied multiple α-MSH fragments — including KPV — for biological activity independent of the parent hormone’s receptor binding profile. KPV can be obtained either through enzymatic cleavage of α-MSH or through direct solid-phase peptide synthesis (SPPS). Research-grade KPV is typically produced via SPPS using Fmoc chemistry, followed by HPLC purification to research-grade purity standards (≥98%). What Does KPV Research Suggest About Inflammatory Pathways? Published research has investigated KPV’s interaction with several molecular components of inflammatory signaling. The most extensively cited body of work comes from intestinal inflammation models, where Dalmasso and colleagues demonstrated that KPV is taken up by intestinal epithelial cells via the PepT1 transporter and reduced markers of inflammation in murine colitis models (Dalmasso et al., 2008 [PubMed]). Subsequent investigations have examined KPV’s interaction with NF-κB signaling and with the production of pro-inflammatory cytokines including IL-6, IL-8, and TNF-α in cell-based assays. Research published in 2025 has additionally examined KPV’s effect on IL-1β production in models of fine-particulate-induced inflammation (Sung et al., 2025). It is important to note that all of these findings are derived from in vitro, ex vivo, and animal model research. Published clinical research in humans is limited. Which Research Areas Have Examined KPV? Intestinal Inflammation Models The 2008 Dalmasso study remains one of the most-cited investigations of KPV. Using both in vitro intestinal epithelial cell cultures and in vivo murine colitis models (DSS-induced and TNBS-induced), researchers reported reductions in inflammatory markers following KPV administration. The study identified PepT1-mediated cellular uptake as a proposed mechanism for KPV’s intracellular activity (Dalmasso et al., 2008 [PubMed]). Skin and Atopic Dermatitis Models KPV has been investigated in dermal research contexts, including iontophoretic transdermal delivery studies and topical formulation research. Pawar and colleagues characterized KPV’s permeation profile through skin models and reported that iontophoresis enhanced peptide delivery compared to passive diffusion (Pawar et al., 2017 [PubMed]). Antimicrobial Activity Studies Research on α-MSH-derived peptides has examined antimicrobial activity against several organisms, including Staphylococcus aureus and Candida albicans, with KPV identified as one of the active fragments retaining a portion of the parent peptide’s antimicrobial profile (Cutuli et al., 2000 [PubMed]; Brzoska et al., 2008 [PubMed]). Fine-Particulate Inflammation Models A 2025 publication examined KPV’s effect on inflammatory markers in cell models exposed to fine particulate matter, with researchers reporting modulation of IL-1β production via proposed antioxidant pathways (Sung et al., 2025). What Is the Reported Mechanism of Action? Across the published literature, three principal mechanisms have been proposed for KPV’s activity in research models: Cellular uptake via PepT1. Intestinal epithelial cells express the PepT1 di/tripeptide transporter, which Dalmasso and colleagues identified as the route by which KPV enters the cytoplasm. Once intracellular, KPV is proposed to interact with downstream signaling components rather than acting via cell-surface receptors (Dalmasso et al., 2008 [PubMed]). NF-κB pathway interaction. Multiple studies have reported that KPV exposure correlates with reduced activation of
Ipamorelin Peptide: A Research Review of Mechanism, Selectivity, and Purity Verification
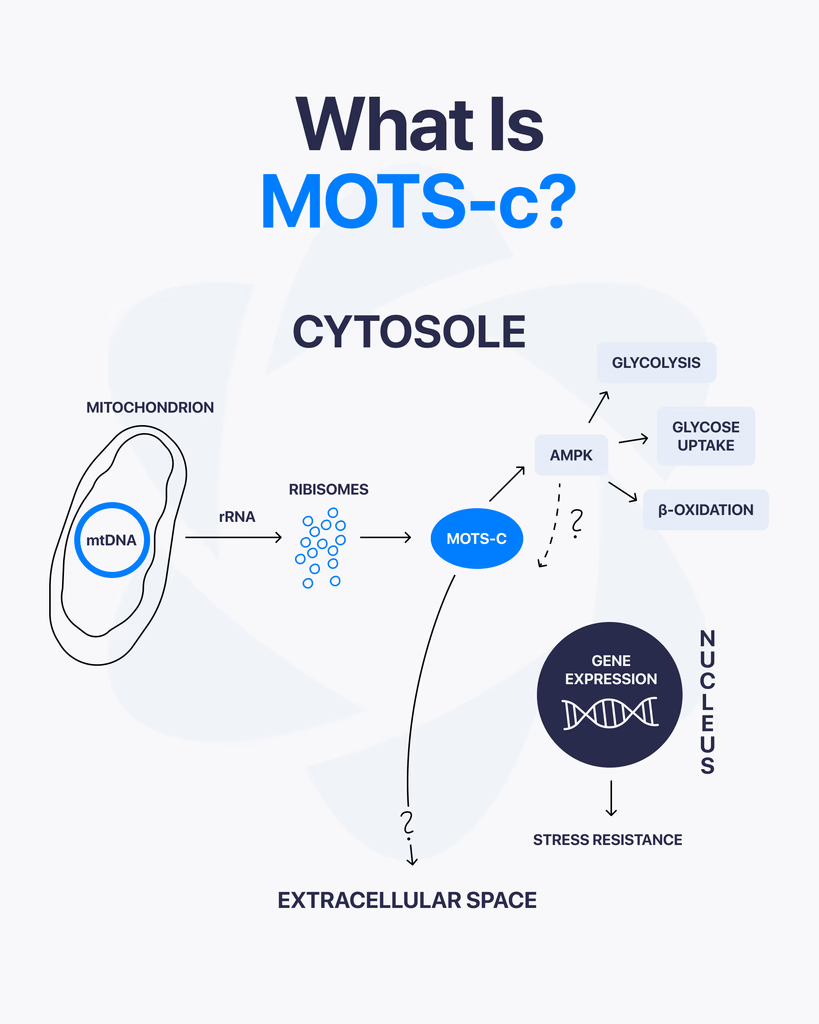
By Dr. Leonard Haberman, Chief Science Officer, OPTMZ Peptides Published March 2, 2026 · Last Updated April 16, 2026 Research Use Only. The information in this article is provided for laboratory research and scientific education. The compounds discussed are not intended for human or animal consumption, clinical use, or therapeutic application. Statements on this site have not been reviewed by the FDA. Ipamorelin is a synthetic pentapeptide studied in endocrine research for its selective stimulation of growth hormone release. Structurally defined as Aib-His-D-2-Nal-D-Phe-Lys-NH₂, it binds the growth hormone secretagogue receptor (GHS-R1a) — the same receptor activated by endogenous ghrelin — and has been characterized in published research as the first GHRP-class ligand with selectivity comparable to GHRH (Raun et al., 1998). This article reviews what the peer-reviewed literature has established about ipamorelin’s mechanism of action, its receptor selectivity, the key preclinical research areas it has been investigated in, and the analytical standards used to verify research-grade ipamorelin purity. What Is Ipamorelin? Ipamorelin is a synthetic five-amino-acid peptide (a pentapeptide) developed in the 1990s as part of research into growth hormone secretagogues — compounds that stimulate endogenous growth hormone (GH) release from the anterior pituitary. It was first characterized by Raun and colleagues at Novo Nordisk in 1998 and described as the first GHRP-receptor agonist whose selectivity for GH release was comparable to that of growth hormone-releasing hormone itself (Raun et al., 1998, PubMed). In current peptide research contexts, ipamorelin is used as a pharmacological tool to investigate GH secretion, GHS-R1a receptor signaling, and related endocrine pathways. It is classified as a research compound and is not approved for clinical therapeutic use in any jurisdiction. What Is the Molecular Structure of Ipamorelin? Ipamorelin’s amino acid sequence is Aib-His-D-2-Nal-D-Phe-Lys-NH₂, where: Aib is α-aminoisobutyric acid, a non-proteinogenic amino acid that introduces conformational rigidity His is L-histidine D-2-Nal is D-2-naphthylalanine, a non-natural D-amino acid D-Phe is D-phenylalanine Lys-NH₂ is C-terminal lysine amide The inclusion of D-amino acids and the unnatural Aib residue is deliberate. These substitutions increase resistance to enzymatic degradation and alter the peptide’s secondary structure, producing a compact, receptor-selective ligand. The molecular formula is C₃₈H₄₉N₉O₅ with a molecular weight of approximately 711.85 g/mol. Structurally, ipamorelin is derived from the GHRP-1 lineage but has been optimized to reduce off-target receptor engagement. This optimization is the basis of the selectivity profile described below. How Does Ipamorelin Stimulate Growth Hormone Release? Ipamorelin is an agonist of the growth hormone secretagogue receptor type 1a (GHS-R1a) — a G-protein-coupled receptor (GPCR) expressed on somatotroph cells of the anterior pituitary and in regions of the hypothalamus. The GHS-R1a is the same receptor that binds endogenous ghrelin. Upon binding, ipamorelin activates the Gq/11 pathway, triggering phospholipase C (PLC) activity, inositol triphosphate (IP₃) generation, and intracellular calcium mobilization. This cascade results in the release of stored growth hormone from somatotroph secretory vesicles into systemic circulation. In pharmacokinetic-pharmacodynamic modeling performed in healthy human volunteers, ipamorelin administration produced a rapid, dose-dependent increase in serum GH, with measurable GH elevation occurring within minutes of subcutaneous or intravenous delivery (Gobburu et al., 1999, PubMed). Unlike synthetic growth hormone itself, which directly supplements circulating GH levels, ipamorelin stimulates the pituitary’s own GH-releasing machinery. Research on GHS-R1a agonists has shown that this mode of action preserves the pulsatile pattern of GH secretion — a pattern considered physiologically important in native endocrine regulation. What Makes Ipamorelin Selective Compared to Other GHRPs? The defining characteristic of ipamorelin in the peer-reviewed literature is its selectivity — specifically, its stimulation of GH release without significant concurrent elevation of adrenocorticotropic hormone (ACTH), cortisol, or prolactin. Earlier growth hormone secretagogues, including GHRP-6 and GHRP-2, reliably stimulate GH release but also produce measurable increases in ACTH and cortisol, and in some studies prolactin as well. This cross-reactivity has historically complicated the use of these earlier peptides as clean research tools for isolating GH-specific effects. Raun et al. (1998) demonstrated in both in vitro and in vivo models that ipamorelin, administered at doses that produced GH responses equivalent to GHRP-6, did not significantly elevate ACTH or cortisol. Johansen et al. (1999) extended these observations and characterized ipamorelin’s receptor binding kinetics, confirming that the selectivity appears to originate from the peptide’s specific binding profile at GHS-R1a and its lack of engagement with receptors responsible for corticotropin release (Johansen et al., 1999, PubMed). For researchers, this matters because it allows the study of GH-dependent pathways with fewer confounding endocrine variables. In bone formation research, for example, Svensson et al. (2000) were able to examine ipamorelin’s influence on bone mineral content in adult rats without simultaneously introducing the catabolic effects of elevated glucocorticoids that would have been expected with less-selective secretagogues (Svensson et al., 2000, PubMed). What Did the Original Discovery Research Establish? Two foundational publications anchor the scientific understanding of ipamorelin. Raun K, Hansen BS, Johansen NL, et al. (1998). Ipamorelin, the first selective growth hormone secretagogue. European Journal of Endocrinology, 139(5):552-561. (PubMed 9849822) — the foundational discovery paper. It established ipamorelin’s potency and efficacy in vitro (using isolated pituitary cells) and in vivo (in swine and rats), benchmarked it against GHRP-6, and documented the distinctive selectivity profile. The paper has been cited more than 180 times in subsequent literature. Johansen PB, Segev Y, Landau D, et al. (1999). Ipamorelin, a new growth-hormone-releasing peptide: Pharmacokinetic and pharmacodynamic properties. (PubMed 10373343) — extended the receptor characterization, established pharmacokinetic parameters in rats, and confirmed in additional models that GH release was not accompanied by ACTH or prolactin elevation. Subsequent research has built on these foundations. Gobburu and colleagues (1999) developed a pharmacokinetic-pharmacodynamic model based on data from human volunteers that described ipamorelin’s GH response curve across a range of doses. The 1998 and 1999 papers remain the primary citations in virtually all contemporary ipamorelin research. How Does Ipamorelin Interact with Somatostatin and Endocrine Feedback? Native GH secretion is regulated by a two-signal system: growth hormone-releasing hormone (GHRH) from the hypothalamus stimulates release, and somatostatin (also hypothalamic) inhibits it. Endogenous ghrelin — and, by extension, GHS-R1a agonists like
Melanotan 2 vs PT-141: A Comparison of Two Melanocortin Agonist Peptides
Melanocortin agonists are a group of compounds studied for how they interact with the melanocortin system, a network of receptors involved in processes like pigmentation, energy balance, and signaling within the body. In peptide research, these compounds are explored to better understand how specific receptor pathways influence different biological responses. What Are Melanocortin Agonists? Melanocortin agonists are substances that activate melanocortin receptors. These receptors are part of a larger system that responds to naturally occurring peptides such as alpha-MSH (melanocyte-stimulating hormone). There are several types of melanocortin receptors, labeled MC1R through MC5R. Each one is linked to different functions within the body. How They Work Here’s the key idea. When a melanocortin agonist binds to a receptor, it triggers a biological response depending on which receptor is activated. For example: MC1R is associated with pigmentation MC3R and MC4R are linked to energy regulation and appetite signaling MC5R is involved in various gland-related functions By targeting these receptors, researchers can study how specific pathways respond under controlled conditions. Common Compounds Studied Several melanocortin-related peptides are frequently explored in research, including: Melanotan I (MT-1) Melanotan II (MT-2) Other synthetic analogs of melanocyte-stimulating hormones These compounds are designed to interact with melanocortin receptors and help researchers observe how activation affects biological systems. Areas of Research Interest Melanocortin agonists are studied across multiple areas, such as: Pigmentation PathwaysResearch often focuses on how receptor activation influences melanin production. Energy Balance and MetabolismSome receptors play a role in regulating appetite and energy use, making them a point of interest in metabolic studies. Cellular SignalingThese compounds help researchers understand how signals are transmitted through receptor pathways and how different systems interact. What Research Indicates Most findings in this field come from preclinical studies and controlled laboratory research. These studies show that melanocortin agonists can influence receptor-specific pathways, but the exact outcomes depend on the compound, dosage, and conditions. Because different receptors produce different responses, the effects are not uniform across all melanocortin agonists. Limitations and Ongoing Research While there is a growing body of research, many questions remain. Most available data comes from: Laboratory experiments Animal studies Early-stage investigations More research is needed to fully understand long-term effects and broader biological implications. Final Thoughts Melanocortin agonists provide a useful way to study how specific receptor systems function. By targeting different melanocortin receptors, researchers can explore pathways related to pigmentation, metabolism, and cellular signaling. At this stage, these compounds remain an active area of research, with ongoing studies continuing to expand what we know.
Peptide Synthesis Explained: SPPS, Purification, and Purity Verification
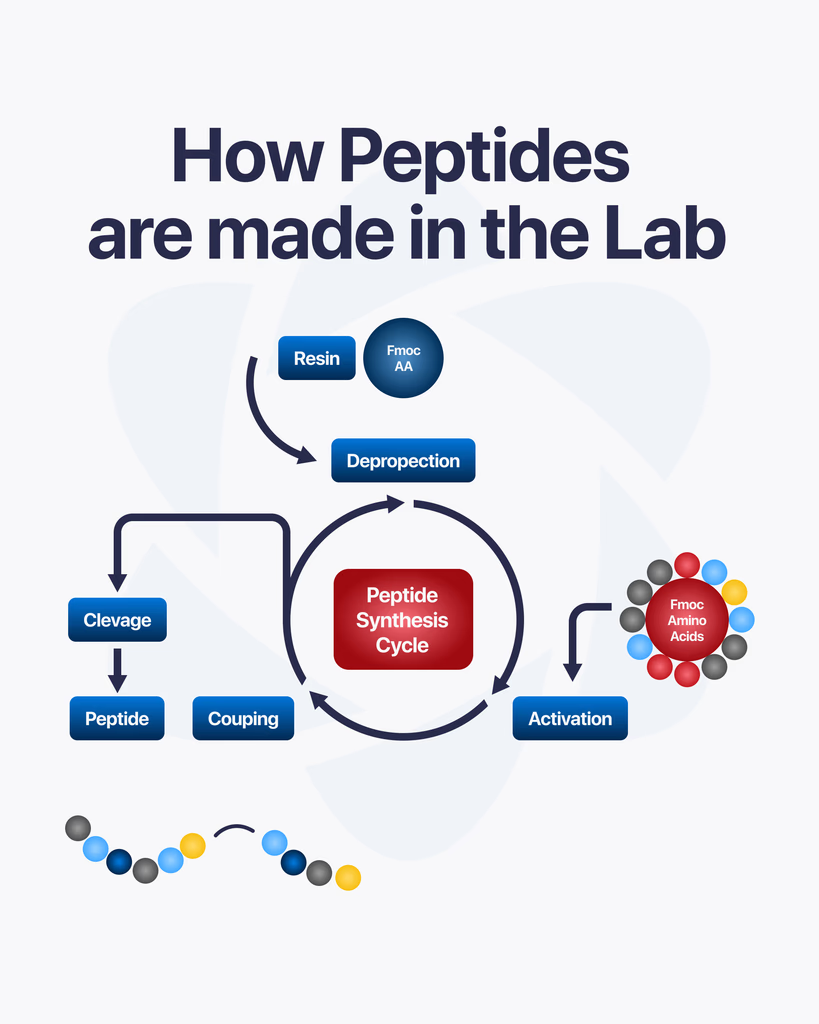
By Dr. Leonard Haberman, Chief Science Officer, OPTMZ Peptides Published: March 2, 2026 | Last Updated: April 16, 2026 Peptide synthesis is the controlled laboratory process by which researchers build peptide chains from individual amino acid subunits. Rather than isolating peptides from biological tissues, synthetic production allows precise sequence specification — making it the standard method for producing research-grade compounds used in pre-clinical and in vitro studies. Understanding how research peptides are synthesized helps researchers make better decisions about supplier selection, purity interpretation, and storage protocols. This article covers the core synthesis method, the purification steps that follow, and the analytical testing that determines whether a finished batch meets research-grade standards. What Is Peptide Synthesis? Peptide synthesis is the process of forming peptide bonds between amino acids in a defined sequence. In biological systems, this occurs through ribosomal translation of mRNA sequences. In laboratory settings, synthesis is performed chemically — allowing researchers to specify any sequence, introduce non-natural amino acids, or produce compounds that do not exist in sufficient quantities in natural sources. The peptide bond itself is a covalent amide linkage between the carboxyl group of one amino acid and the amino group of the next. Each addition extends the chain by one residue. A ten-residue peptide requires nine successful bond formations, each carried out under controlled conditions (Merrifield, 1963 — PubMed). The precision of the final sequence determines the compound’s research relevance. A single incorrect residue, deleted residue, or epimeric impurity can alter receptor binding, biological stability, or assay behavior. This is why synthesis methodology and post-synthesis verification are inseparable from research utility. What Is Solid-Phase Peptide Synthesis (SPPS)? Solid-phase peptide synthesis (SPPS) is the dominant method for producing research-grade peptides. Introduced by Robert Bruce Merrifield in 1963, the technique anchors the growing peptide chain to an insoluble polymer resin, which allows excess reagents and byproducts to be washed away between each coupling step (Merrifield, 1963 — PubMed). How Does SPPS Work? The synthesis proceeds through a repeating cycle: 1. Resin loading. The C-terminal amino acid of the target sequence is attached to the solid resin support via a linker. The resin physically anchors the chain throughout synthesis, enabling filtration-based purification between steps. 2. Deprotection. Amino acids carry protecting groups on their reactive side chains and N-terminus. Before each coupling step, the N-terminal protecting group is removed — a process called deprotection — exposing the free amino group for the next reaction. 3. Coupling. The next amino acid in the sequence is added in excess with a coupling reagent. The coupling reagent activates the incoming amino acid’s carboxyl group, enabling it to react with the deprotected N-terminus on the resin-bound chain. This forms a new peptide bond. 4. Washing. Excess reagents, activating agents, and byproducts are washed away with solvent. The resin retains the growing peptide chain. 5. Iteration. The deprotection → coupling → washing cycle repeats for each amino acid in the sequence, proceeding from C-terminus to N-terminus. 6. Cleavage and global deprotection. When the full sequence has been assembled, the completed peptide is cleaved from the resin. Simultaneously, remaining side-chain protecting groups are removed, yielding the free peptide in solution. The primary advantage of SPPS over solution-phase synthesis is the ability to use large excesses of coupling reagents to drive reactions to completion — a requirement when even a small percentage of incomplete couplings results in deletion sequences or truncated impurities that reduce the final purity. Two main SPPS strategies exist based on the N-terminal protecting group chemistry used: Boc (t-Boc) SPPS and Fmoc SPPS. Fmoc chemistry has become the standard for most research peptide synthesis due to its milder deprotection conditions and compatibility with a wider range of side-chain protecting groups (Carpino and Han, 1972 — PubMed). Why Do Incomplete Coupling Reactions Matter for Research? Each coupling step in SPPS does not achieve 100% efficiency. A typical coupling reaction yields 98–99.9% efficiency depending on the amino acid, the sequence context, and the coupling reagent used. Across a long sequence, these small inefficiencies accumulate. For a 20-residue peptide synthesized at 99% per-step coupling efficiency, the theoretical yield of the fully correct sequence is approximately 82% (0.99²⁰). The remaining fraction consists of deletion peptides — sequences where one or more amino acids have been skipped — and truncated sequences that terminated early. These impurities do not always share the same biological behavior as the target peptide. In research applications where receptor selectivity, binding affinity, or downstream signaling effects are being studied, impurity profiles that deviate significantly from the intended sequence can introduce confounding variables into experimental results (Chan and White, 2000 — PubMed). This is why purification is not optional for research-grade compounds. How Is a Research Peptide Purified After Synthesis? The crude peptide cleaved from the resin contains the target compound alongside synthesis byproducts, incomplete sequences, and protecting group fragments. Purification removes these impurities to yield a product that meets defined purity thresholds for research use. Reversed-Phase High-Performance Liquid Chromatography (RP-HPLC) Reversed-phase HPLC (RP-HPLC) is the standard purification method for research peptides. The crude peptide mixture is dissolved and injected onto an HPLC column packed with a hydrophobic stationary phase (typically C18). As a mobile phase gradient of water and organic solvent (usually acetonitrile) moves through the column, peptide components separate based on their hydrophobicity. The target peptide elutes at a characteristic retention time, and the corresponding fraction is collected. Other fractions — containing deletion sequences, oxidation products, and protecting group adducts — are discarded. Preparative RP-HPLC can achieve purity levels exceeding 98% for most research peptide sequences. The resulting material is lyophilized (freeze-dried) to produce a stable white to off-white powder suitable for storage and research use. How Is Peptide Purity Analytically Verified? Purification produces a compound of defined purity. Analytical testing confirms that purity and verifies the compound’s identity. For research-grade peptides, this is not a quality assurance formality — it is the primary evidence that the compound in the vial is what the label states. What Does HPLC Purity Testing Measure? Analytical
Selank Peptide: Research Overview, Mechanisms, and Pre-Clinical Study Findings

Selank is a synthetic peptide that has been studied for its potential effects on the nervous system. It’s derived from a naturally occurring peptide involved in immune signaling, but modified to improve stability and activity in research settings. Over time, it has gained attention for how it may interact with brain function, particularly in areas related to mood and cognitive processes. What Is Selank? Selank is a short peptide analog based on tuftsin, a naturally occurring compound linked to immune system activity. Researchers have explored how this modified version behaves in the body, especially in relation to neurological pathways. Because of its structure, Selank is often studied for its potential to influence signaling in the brain without acting like traditional compounds. How It’s Studied in Research In laboratory settings, Selank is examined for how it interacts with neurotransmitters and brain receptors. Some studies suggest it may affect systems involving: GABA (gamma-aminobutyric acid) Serotonin pathways Dopamine balance These systems are all closely tied to mood regulation, stress response, and cognitive function. Potential Areas of Interest Researchers have explored several areas where Selank may play a role: Cognitive FunctionSome studies suggest it may support memory and learning processes by influencing how signals are transmitted in the brain. Stress and Anxiety ResponseThere is interest in how Selank interacts with stress-related pathways, particularly through its effect on neurotransmitter balance. Immune System InteractionSince it is derived from tuftsin, Selank is also studied for how it may influence immune signaling and inflammatory responses. What Current Research Suggests Most findings around Selank come from preclinical and early-stage studies. These suggest that the peptide may have regulatory effects on certain brain functions without producing strong sedative or stimulant effects. However, it’s important to understand that research is still developing. There is limited large-scale clinical data, and many findings are based on controlled laboratory environments. Limitations and Ongoing Study Here’s the honest picture. While early research is promising, there isn’t enough conclusive evidence to fully define Selank’s role across all potential areas. Most studies focus on: Animal models Cellular-level analysis Small-scale research trials Further investigation is needed to better understand long-term effects and broader applications. Final Thoughts Selank is an interesting subject within peptide research, especially when it comes to studying brain function and signaling pathways. Its interaction with neurotransmitters and immune-related processes makes it a focus for ongoing research. For now, it remains a compound being actively studied rather than a fully established solution in any specific area.
How to Reconstitute Lyophilized Peptides for Research Use

By Dr. Leonard Haberman, Chief Science Officer, OPTMZ Peptides Published: March 2, 2026 | Last Updated: April 15, 2026 Lyophilized peptides are supplied as a dry, powdered solid specifically because the freeze-drying process extends compound stability and preserves structural integrity during storage and transit. Before a lyophilized peptide can be used in laboratory research, it must be reconstituted — dissolved into an appropriate solvent at a known concentration. Performing this step correctly determines the accuracy and reproducibility of downstream experiments. This guide covers what lyophilization does to peptide compounds at the molecular level, how to select the right reconstitution solvent, step-by-step reconstitution procedure, concentration calculations, and storage protocols for reconstituted research peptide solutions. What Is Lyophilization and Why Does It Matter for Research Peptides? Lyophilization — commonly called freeze-drying — is a dehydration process that removes water from a compound through sublimation rather than evaporation. The sample is first frozen, then placed under vacuum, causing ice to transition directly from solid to vapor without passing through a liquid phase. The result is a stable, dry solid that retains the compound’s molecular structure. For research peptides, lyophilization addresses a fundamental stability problem. Peptides in aqueous solution are susceptible to hydrolysis, oxidation, and microbial degradation. Studies examining peptide stability in solution have documented significant degradation within days to weeks under non-optimal conditions, depending on the compound’s sequence, pH sensitivity, and temperature exposure (Manning et al., 2010 — PubMed). The lyophilized state substantially reduces these degradation pathways by removing the water molecules that facilitate them. The Three-Stage Lyophilization Process Stage 1 — Freezing: The peptide solution is rapidly cooled, typically to between -40°C and -80°C. Rapid freezing creates smaller ice crystals, which reduces structural disruption to the peptide matrix. The rate of freezing affects the final product’s physical characteristics and reconstitution behavior. Stage 2 — Primary Drying (Sublimation): The chamber pressure is reduced below the water vapor pressure, and shelf temperature is gradually raised. Frozen water sublimes directly to vapor and is collected by a condenser. This stage removes approximately 95% of the water content and accounts for the majority of the lyophilization cycle time. Stage 3 — Secondary Drying (Desorption): The remaining bound water — water molecules adsorbed to the peptide surface rather than frozen — is removed by further increasing shelf temperature at low pressure. Residual moisture content in pharmaceutical-grade lyophilized compounds is typically targeted at 1–3% by weight. Residual moisture above this threshold accelerates degradation even in the dry state (Abdul-Fattah et al., 2007 — PubMed). The final product is a lyophilized “cake” or powder in a sealed vial, suitable for long-term storage and shipment at ambient temperature when properly sealed. Why the Research-Grade Supply Standard Matters Here The purity and structural integrity of a lyophilized peptide is established at the time of manufacture and confirmed by batch testing — not at reconstitution. A compound that tests at 99.1% purity by HPLC in the lyophilized state will not improve during reconstitution. Conversely, a compound with an undisclosed impurity profile will carry those impurities into every experiment conducted with it. This is why batch-specific certificate of analysis data is the only reliable baseline for research work. Each OPTMZ Peptides batch is independently verified by Krause Analytical (DEA-registered, ISO/IEC 17025-certified, Austin TX) using HPLC purity analysis, mass spectrometry identity confirmation, endotoxin testing, heavy metals screening by ICP-MS, microbial testing, pH stability, and visual inspection. Published COA results for all batches are searchable in the COA Vault. Selecting the Right Reconstitution Solvent Solvent selection is among the most consequential decisions in peptide reconstitution. The wrong solvent can cause aggregation, precipitation, or structural modification that renders the solution unusable. Bacteriostatic Water (BAC Water) is the standard reconstitution solvent for most research peptides. It contains 0.9% benzyl alcohol, which inhibits microbial growth and extends the usability of the reconstituted solution — typically 28–30 days when stored at 2–8°C. BAC water is appropriate for the majority of peptides in the research catalog, including BPC-157, TB-500, CJC-1295, Ipamorelin, Sermorelin, and others. OPTMZ Peptides supplies Bacteriostatic Water 30mL for this purpose. Acetic Acid 0.6% (dilute acetic acid) is used as a reconstitution solvent for peptides that exhibit poor solubility in neutral aqueous solutions. Growth hormone releasing peptides and some longer-chain peptides may require acetic acid as an initial solvent before dilution with BAC water. Sterile Water is acceptable for immediate-use applications where the reconstituted solution will be used within 24 hours. It offers no antimicrobial protection and is not suitable for multi-day storage of reconstituted peptides. DMSO (dimethyl sulfoxide) is appropriate for hydrophobic peptides that are insoluble in aqueous solutions. DMSO solutions are typically diluted with aqueous buffer immediately before use. Its use is more common in in-vitro cell culture applications than in other research contexts. When solubility is uncertain, consult the compound’s published literature or the batch COA notes. Poorly soluble peptides may require sonication (brief low-power ultrasound treatment) or gentle heating to facilitate dissolution — never vortex vigorously, as mechanical shear can disrupt peptide conformation. Step-by-Step Reconstitution Protocol The following procedure applies to standard lyophilized peptide vials for laboratory research use. Materials required: Lyophilized peptide vial (labeled with batch number — confirm against COA Vault) Reconstitution solvent (BAC Water or appropriate alternative) Sterile 1mL or 3mL syringe 18–23 gauge needle for solvent withdrawal Sterile swabs or alcohol wipes Procedure: Equilibrate the vial to room temperature before opening or injecting solvent. Temperature differentials between the cold vial and room-temperature solvent can cause pressure differentials and solution splatter. Allow 10–15 minutes at room temperature. Swab the vial septum with a sterile alcohol wipe. Allow to air dry. Calculate the volume of solvent required for the desired concentration (see Concentration Calculation section below) and draw this volume into the syringe. Inject the solvent slowly against the inside wall of the vial — do not inject directly onto the lyophilized cake. Directing the stream along the glass wall allows gentle dissolution without disrupting the peptide matrix. Gently swirl — do not vortex. Allow the vial
Amino Acid Chain, Peptide Bonds, and Proteins: Structural Differences in Research Context

If you’re working around peptide research, you’ll keep seeing these three terms: amino acids, peptides, and proteins. They’re closely connected, but not the same thing. Understanding how they relate makes everything else easier to follow. Starting Point: Amino Acids Amino acids are the smallest units in this chain. They’re the basic building blocks that make up both peptides and proteins. Each amino acid has a simple structure, but when they link together, they form more complex compounds. There are many types, and the specific sequence in which they connect determines the final structure and function. In short, amino acids are the foundation. What Are Peptides? Peptides are short chains of amino acids linked together through peptide bonds. You can think of them as the middle layer between single amino acids and full proteins. They usually contain a limited number of amino acids, often ranging from just a few to a few dozen. Because of their smaller size, peptides are often easier to study and analyze in research settings. Peptides are involved in many biological processes, including signaling and regulation. What Are Proteins? Proteins are much larger and more complex structures made from long chains of amino acids. These chains fold into specific shapes, which determine how the protein functions. Unlike peptides, proteins can contain hundreds or even thousands of amino acids. This complexity allows them to perform a wide range of functions, from structural support to enzymatic activity. The Key Differences Here’s the simplest way to break it down: Amino acids are individual building blocks Peptides are short chains of amino acids Proteins are long, fully structured chains with complex functions The difference mainly comes down to size, structure, and biological role. Why This Matters in Research In peptide research, knowing where a compound fits in this chain helps researchers understand how it behaves. For example: Amino acids are studied for their chemical properties Peptides are often used to explore signaling and targeted interactions Proteins are analyzed for their broader biological functions Each level offers different insights depending on the goal of the research. Final Thoughts Amino acids, peptides, and proteins are all part of the same biological system, just at different levels of complexity. Once you understand how they connect, it becomes much easier to navigate research and terminology in this field.
HPLC Peptide Testing: How Research-Grade Purity Is Measured and Verified

By Dr. Leonard Haberman, Chief Science Officer, OPTMZ Peptides Published: March 2, 2026 | Last Updated: April 15, 2026 High-performance liquid chromatography (HPLC) is the primary analytical method used to measure peptide purity in research-grade compounds. When a Certificate of Analysis (COA) reports a purity percentage for a research peptide, that number almost always comes from HPLC data. Understanding what HPLC measures — and what it cannot — is essential context for any researcher evaluating compound quality. What Does HPLC Actually Measure in a Peptide Sample? HPLC does not test a peptide’s biological activity. It measures the compositional purity of a sample — specifically, what percentage of the detectable material in a vial corresponds to the target peptide versus other compounds. In reverse-phase HPLC (RP-HPLC), the method used for most peptide analysis, a liquid sample is pushed under high pressure through a column packed with hydrophobic stationary phase material. Compounds in the sample travel through the column at different rates depending on how strongly they interact with the stationary phase. The detector records a signal as each compound exits, producing a chromatogram: a graph of detector response over time. Each peak in a chromatogram represents a distinct compound. The target peptide produces the largest peak. Impurities, synthesis byproducts, degradation products, or residual reagents produce smaller secondary peaks. Purity is calculated as the area of the target peak divided by the total area of all detected peaks, expressed as a percentage. A sample reporting 99.2% purity means that 99.2% of the detectable material in that vial is the target peptide. The remaining 0.8% comprises everything else the detector can resolve. Why Reverse-Phase HPLC Is the Standard for Peptide Research Three properties of RP-HPLC make it the preferred method for research peptide analysis: Sensitivity to hydrophobic differences. Peptides vary in hydrophobicity depending on their amino acid sequence. RP-HPLC separates compounds based on these differences with sufficient resolution to distinguish a target peptide from closely related impurities, including truncated sequences, deletion sequences, or oxidation products. Studies validating RP-HPLC for peptide purity assessment have confirmed its suitability for detecting impurities at sub-1% levels (Mant & Hodges, 1991, Journal of Chromatography A PubMed). Reproducibility. Properly calibrated RP-HPLC systems generate consistent results across repeated injections of the same sample, making it a reliable basis for batch-to-batch comparison. Compatibility with standard COA reporting. HPLC purity values expressed as area-under-curve percentages are the accepted format for research-grade compound certificates of analysis. This standardization allows researchers to compare purity data across vendors using a consistent metric. For research applications where compound consistency directly affects experimental outcomes, RP-HPLC purity data is the minimum acceptable quality documentation. Research examining peptide stability has noted that purity degradation as small as 2–3% can introduce measurable variability in in vitro assay results (Bhatt et al., 2011, International Journal of Peptides PubMed). How to Read an HPLC Chromatogram on a Peptide COA When reviewing a COA from OPTMZ Peptides, the “Lab Analysis” tab provides the HPLC result for that specific batch. Here is how to interpret the key components: The main peak retention time. This is the time point at which the target peptide elutes from the column. Retention time is characteristic of the compound’s structure. A shift in retention time between batches can indicate a structural difference. The peak area percentage. This is the purity figure. On OPTMZ COAs, the minimum accepted purity is 98.0% — batches testing below this threshold are rejected before entering inventory. Most batches test between 98.5% and 99.9%. Secondary peaks. Minor peaks are expected and do not indicate a failed batch unless they exceed acceptable limits. The identity of secondary peaks requires mass spectrometry — HPLC alone can detect them but cannot confirm what they are. The baseline. A flat, stable baseline with clearly resolved peaks indicates a clean, well-executed analysis. An unstable or noisy baseline may indicate column degradation or sample preparation issues; it does not necessarily invalidate the result but warrants scrutiny. For a direct example: In Batch #BPC-2026-03-15, HPLC analysis of BPC-157 returned 99.4% purity as measured by Krause Analytical. The chromatogram showed a single dominant peak at the expected retention time with no secondary peaks exceeding 0.2% area. The full COA for this batch is available in the OPTMZ COA Vault. What HPLC Cannot Confirm Alone HPLC purity data answers one question: how much of the sample is the target compound. It does not answer: Identity. A sample could theoretically achieve a high HPLC purity figure while being the wrong compound. Identity confirmation requires mass spectrometry (MS), which verifies the molecular weight and fragmentation pattern of the compound. HPLC and MS are used together on every OPTMZ batch for this reason. Endotoxin levels. Bacterial endotoxins are not detectable by HPLC. Endotoxin testing (LAL method) is a separate required test for research-grade peptides. Residual solvents or heavy metals. These contaminants require dedicated analytical methods — ICP-MS for metals, headspace GC for residual solvents. A COA that reports only HPLC purity is incomplete for research purposes. OPTMZ’s standard testing panel covers seven methods: HPLC (purity), mass spectrometry (identity), endotoxin (LAL), heavy metals (ICP-MS), microbial, pH stability, and visual inspection — all conducted by Krause Analytical, a DEA-registered, ISO/IEC 17025-certified laboratory in Austin, TX. Batch-to-Batch Consistency: Why Single-Batch HPLC Data Is Insufficient A single high-purity result does not establish that a supplier’s process is consistent. Research settings require reproducible inputs: a compound that tests at 99.1% in one batch but 96.0% in the next introduces an uncontrolled variable into any experimental protocol. Evaluating a vendor’s HPLC history across multiple batches is the only reliable way to assess process consistency. The OPTMZ COA Vault maintains a searchable archive of published COA results by batch number — accessible directly from the vial label. Researchers can verify the purity of the specific batch in hand, compare it against prior batches, and assess whether the supplier’s process is stable over time. No competitor publishes a historical COA archive with batch-level searchability. HPLC Purity as a
Lyophilized Peptides: What They Are, How Excipients Work, and Storage Guidelines for Research
When working with peptides, most of the attention goes to the active compound itself. But there’s another important component that often gets overlooked, excipients. These supporting substances play a key role in how peptides are stored, handled, and studied. What Are Excipients? Excipients are inactive substances added alongside a peptide. They don’t produce biological effects on their own, but they help maintain the stability and usability of the peptide during storage and research. Think of them as support agents. Their job is to protect the peptide, improve consistency, and make handling easier in laboratory settings. Why Excipients Are Used in Peptide Research Peptides can be sensitive to environmental conditions like temperature, moisture, and light. Without proper support, they may degrade or lose effectiveness. Excipients are used to: Stabilize peptide structure Extend shelf life Prevent degradation during storage Improve solubility for reconstitution Ensure consistent handling across batches This makes them essential for maintaining reliability in research work. Common Types of Excipients Different excipients are selected depending on the peptide and how it will be used in research. Some of the most common include: Bulking AgentsThese help give the peptide substance physical volume, especially in lyophilized (freeze-dried) form. Mannitol is a commonly used example. StabilizersStabilizers protect peptides from breaking down over time. They help maintain the molecular structure during storage and transport. BuffersBuffers control pH levels, which is important because peptides can be sensitive to even small changes in acidity or alkalinity. SolubilizersThese help peptides dissolve properly when being prepared for research use, ensuring accurate concentration and handling. How Excipients Affect Research Quality Even though excipients are inactive, they still influence how a peptide performs in a research environment. Poor formulation can lead to instability, inaccurate dosing, or inconsistent results. That’s why high-quality peptide preparation always includes carefully selected excipients that match the compound’s characteristics. Things to Keep in Mind Not all excipients are the same, and their compatibility with specific peptides matters. Researchers often consider: The chemical stability of the peptide Storage conditions and shelf life Reconstitution requirements Interaction between excipients and the peptide Choosing the right combination is part of ensuring reliable and repeatable outcomes. Final Thoughts Excipients may not be the main focus in peptide research, but they play a critical supporting role. From stabilizing the compound to improving handling and storage, they help ensure that peptides remain consistent and usable throughout the research process. Ignoring excipients can lead to compromised results, while the right formulation helps maintain accuracy and confidence in every study.