By Dr. Leonard Haberman, Chief Science Officer, OPTMZ Peptides — Updated April 17, 2026
Research Use Only. All OPTMZ Peptides products and all content on this site are provided strictly for in-vitro laboratory research conducted by qualified professionals. These compounds are not intended for human or animal dosing, injection, or ingestion. The statements on this page have not been reviewed by the FDA.
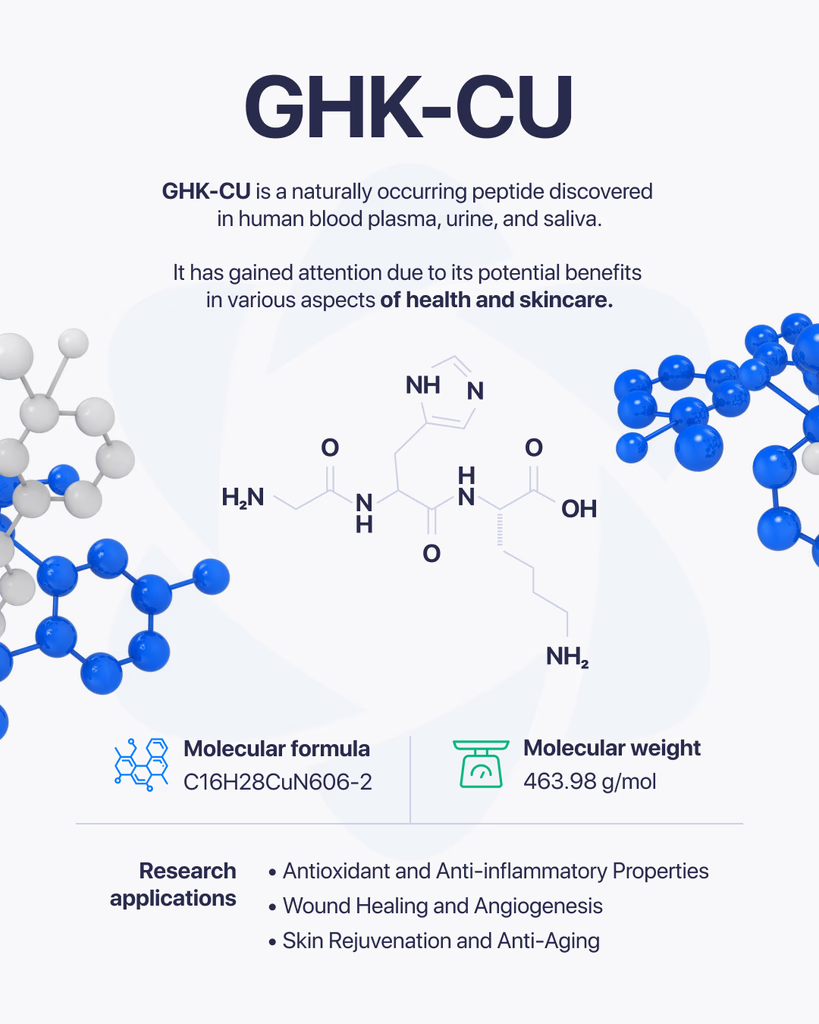
GHK-Cu is a copper-binding tripeptide composed of glycine, histidine, and lysine (Gly-His-Lys) complexed with a divalent copper ion (Cu²⁺). First isolated from human plasma in 1973, the peptide has been studied for decades across wound healing, cellular signaling, gene expression, and extracellular matrix research. This article summarizes what peer-reviewed research has examined about GHK-Cu, how the copper coordination chemistry is characterized analytically, and how research-grade GHK-Cu is verified by independent third-party laboratories prior to distribution to investigators.
What Is GHK-Cu?
GHK-Cu refers to the copper complex formed when the tripeptide glycyl-L-histidyl-L-lysine (GHK) coordinates with a Cu²⁺ ion. The free tripeptide was first isolated from human plasma by Loren Pickart in 1973, who observed that plasma from younger donors contained higher concentrations of the tripeptide than plasma from older donors (Pickart, 2008 PMID: 18644225).
The structural properties of interest in a research context include:
Amino acid sequence: Glycine — L-Histidine — L-Lysine
Molecular formula (peptide): C₁₄H₂₄N₆O₄
Peptide molecular weight: 340.39 g/mol
Copper-bound complex molecular weight: 402.9 g/mol (approximate, with Cu²⁺ coordination)
CAS number (GHK-Cu): 49557-75-7
Coordination chemistry: The imidazole nitrogen of histidine, the α-amino group of glycine, and the deprotonated amide nitrogen between glycine and histidine form a planar coordination environment around the Cu²⁺ center, with lysine residues projecting outward.
This coordination geometry is what distinguishes GHK-Cu from the uncomplexed tripeptide. Research has examined the copper complex as an analytically distinct species from free GHK, with different absorption characteristics, different solubility behavior, and different affinity for cellular receptors and matrix components (Pickart & Margolina, 2018 PMID: 29986520).
How Does GHK-Cu Work in Research Models?
Research published over the past five decades has investigated multiple pathways through which GHK-Cu has been shown to act in pre-clinical and in-vitro systems. The mechanism of interest is multifactorial and is typically described in the literature across four categories.
1. Copper Ion Transport
In cell culture and cell-free systems, the GHK carrier has been studied for its role in shuttling Cu²⁺ ions into cells and presenting copper to cuproenzymes such as lysyl oxidase and superoxide dismutase. Researchers have characterized this as a physiological copper-delivery function rather than a pharmacological one (Pickart, 2008 PMID: 18644225).
2. Modulation of Gene Expression
Gene expression profiling studies have examined GHK-Cu’s interaction with transcriptional programs in fibroblast and keratinocyte models. In one of the most widely cited datasets, GHK was reported to modulate the expression of more than 4,000 genes in human fibroblasts, affecting pathways related to extracellular matrix assembly, antioxidant defense, and cellular stress response (Pickart, Vasquez-Soltero & Margolina, 2015 PMID: 26236730).
3. Extracellular Matrix Research
In fibroblast cultures, the GHK-Cu complex has been studied for its effects on type I collagen synthesis and matrix remodeling. The original Maquart et al. (1988) study reported increased collagen synthesis in fibroblast cultures treated with the tripeptide-copper complex, establishing the basis for subsequent extracellular matrix research (Maquart et al., 1988 PMID: 3169264).
4. Antioxidant and Cellular Stress Research
In-vitro research has examined GHK-Cu’s interactions with reactive oxygen species and its potential to chelate redox-active metal ions. These studies are conducted in cell-free and cell-culture systems and do not constitute evidence of any effect on human subjects (Pickart & Margolina, 2018 PMC8789089).
It is important to note that the overwhelming majority of published GHK-Cu findings derive from in-vitro, ex-vivo, or animal-model research. Human clinical data is limited and does not support therapeutic claims.
Why the Copper Component Matters Analytically
GHK as a free tripeptide and GHK-Cu as a copper complex are different analytical species. For research contexts where a study protocol specifies the copper complex, verifying that the supplied compound is in fact the complexed form — not the free tripeptide — is a methodological requirement.
Analytically, the copper complex can be distinguished from free GHK through:
UV-Vis spectroscopy: GHK-Cu has a characteristic d-d transition absorbance near 525 nm, absent in the uncomplexed peptide.
HPLC retention behavior: The copper complex elutes differently from the free tripeptide on reverse-phase C18 columns.
Mass spectrometry: The mass shift corresponding to Cu²⁺ incorporation is directly observable.
A supplier that does not publish HPLC and mass spectrometry verification for a GHK-Cu batch cannot confirm to a researcher that the vial contents are the complexed form at the labeled stoichiometry. This is one of the reasons published, batch-level COAs are a meaningful quality signal in peptide research procurement.
Verifying Research-Grade GHK-Cu Purity
Research-grade GHK-Cu verification at OPTMZ Peptides is conducted through a third-party laboratory — Krause Analytical, a DEA-registered, ISO/IEC 17025-certified laboratory based in Austin, Texas. Every batch is tested across the following methods before inventory release:
Reverse-phase HPLC — purity quantification. Batches below 98% are rejected.
Mass spectrometry — identity confirmation, including verification of the copper-complexed species.
Endotoxin (LAL) — bacterial endotoxin quantification.
Heavy metals (ICP-MS) — detection of non-specified metal contamination.
Microbial testing — total aerobic and yeast/mold counts.
pH stability — reconstitution behavior.
Visual inspection — lyophilized cake integrity, color, and reconstitution clarity.
Typical GHK-Cu batch results fall in the 98.5–99.9% purity range by HPLC. Batch-specific results are published in the OPTMZ COA Vault, searchable by the batch number printed on each vial.
Reading a GHK-Cu Certificate of Analysis
A complete GHK-Cu COA from an ISO/IEC 17025 laboratory includes:
Batch number (matched to the vial label)
Test date and release date
HPLC purity percentage with chromatogram
Mass spectrometry identity confirmation with expected vs. observed m/z
Endotoxin result (EU/mg)
Heavy metals panel (Pb, As, Cd, Hg by ICP-MS)
Microbial results
Testing laboratory name, address, and accreditation number
Analyst signature and laboratory stamp
A COA without a named testing laboratory, without a batch number, or without a chromatogram is not a research-grade COA — it is a marketing asset. Researchers evaluating peptide suppliers should be able to cross-reference the batch number on a received vial directly to a published COA document.
Research-Grade vs. Cosmetic-Grade GHK-Cu
GHK-Cu is sold across two distinct market classifications that should not be conflated:
Cosmetic-grade GHK-Cu is formulated into topical preparations (serums, creams) and is typically supplied at lower purity thresholds and without research-use documentation. It is regulated as a cosmetic ingredient.
Research-grade GHK-Cu is supplied as a lyophilized powder, intended exclusively for in-vitro laboratory research by qualified professionals. Research-grade material is accompanied by a Certificate of Analysis and has been tested for purity, identity, and contamination panels appropriate to research use.
OPTMZ Peptides supplies research-grade material only. It is not a cosmetic ingredient supplier, and its GHK-Cu product listing is intended for qualified researchers and authorized professionals conducting in-vitro work. Research-grade GHK-Cu is also incorporated into OPTMZ’s GLOW research blend (BPC-157 + GHK-Cu + TB-500) and KLOW research blend (BPC-157 + GHK-Cu + TB-500 + KPV), both supplied with batch-level COA documentation.
Stability and Handling in Research Settings
Lyophilized GHK-Cu, when stored under appropriate conditions, has been reported in the analytical literature to retain purity over extended timeframes. Standard research handling conditions are:
Unopened lyophilized vial: store at −20 °C, protected from light.
Reconstituted material: store at 2–8 °C; typical research stability window of 14–30 days in bacteriostatic water, depending on concentration and reconstitution solvent.
Reconstitution solvent: bacteriostatic water (0.9% benzyl alcohol) is standard for research protocols; sterile water for injection is used where preservative-free reconstitution is required.
Freeze-thaw cycles: minimize. Repeated freeze-thaw has been documented to accelerate purity loss in peptide solutions.
Because copper-complexed peptides are light- and oxidation-sensitive, reconstituted GHK-Cu should be stored in amber or light-protected vials where possible, and solutions that have developed visible discoloration should be re-characterized by HPLC before use in research protocols.
How OPTMZ Peptides Verifies Every GHK-Cu Batch
OPTMZ Peptides operates on a Verified-First model: no claim on the site is made that is not backed by published, downloadable, batch-level laboratory data. For GHK-Cu specifically, this means:
Every inventory batch is tested by Krause Analytical prior to release. Nothing below 98% purity enters inventory.
The Certificate of Analysis for each batch is published at the COA Vault, searchable by batch number, before the batch is listed for order.
The testing methodology is documented publicly on the How We Test page.
Historical COAs remain available after a batch is exhausted — the archive compounds over time and functions as a queryable data record of supply quality.
OPTMZ additionally operates a third-party testing service, accepting samples from researchers who wish to independently verify peptides obtained from other suppliers.
For researchers evaluating whether to incorporate a given supplier’s GHK-Cu into a published study, the ability to directly reference a batch number to a laboratory-issued COA is the primary quality signal that distinguishes verified research-grade material from unverified supply.
Dr. Leonard Haberman is Chief Science Officer at OPTMZ Peptides, overseeing analytical quality assurance and third-party laboratory partnerships with a focus on HPLC-based purity verification and research-grade peptide compound validation. All research peptides sold by OPTMZ Peptides are intended strictly for laboratory research use only.