What Is Tesamorelin? A Research Overview
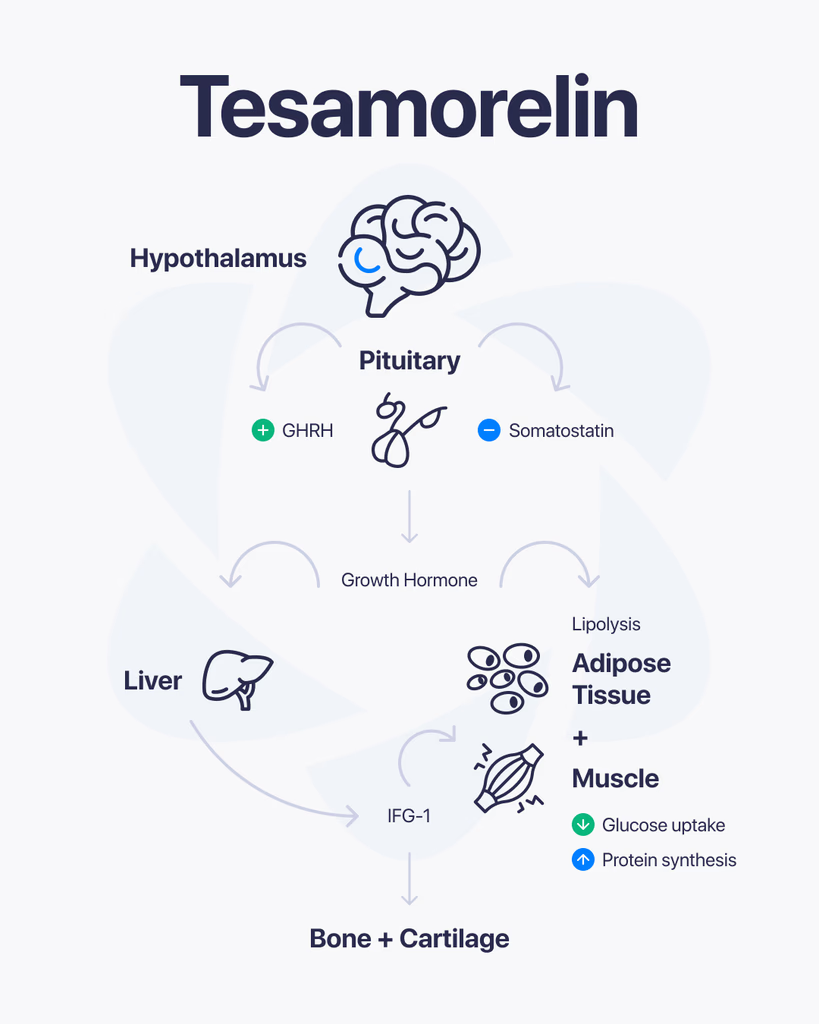
Tesamorelin is a synthetic peptide studied for its role in stimulating the release of growth hormone. It belongs to a class of compounds designed to mimic natural hormone signals rather than replace them directly. Because of this mechanism, it has become a subject of interest in research focused on hormone regulation and metabolic processes. What Is Tesamorelin? Tesamorelin is an analog of growth hormone–releasing hormone (GHRH). Its primary function in research is to signal the pituitary gland to produce and release growth hormone. Instead of introducing growth hormone from the outside, it works by activating the body’s own production system. How It Works Here’s the basic idea. Tesamorelin binds to receptors that trigger the release of growth hormone. This leads to an increase in natural GH levels, which then interact with various tissues in the body. Researchers study this process to understand how hormone signaling affects different biological functions. Why It’s Studied Tesamorelin is explored because it provides a way to study growth hormone regulation through natural pathways. Some key areas of interest include: Hormone release patterns Metabolic signaling Interaction between GH and different tissues Regulation of energy balance By focusing on signaling rather than direct hormone replacement, researchers can observe how the system behaves under controlled stimulation. Areas of Research Focus Growth Hormone RegulationUnderstanding how GH is released and controlled within the body. Metabolic ActivityStudying how hormone signaling affects energy use and processing. Cellular Response MechanismsAnalyzing how tissues respond to changes in hormone levels. What Research Suggests So Far Most available data comes from controlled studies and early-stage research. These indicate that Tesamorelin can effectively stimulate growth hormone release through GHRH pathways. However, outcomes vary depending on study conditions such as dosage, timing, and experimental design. Limitations and Current Understanding Here’s the honest take. While Tesamorelin has been studied in various contexts, there is still ongoing research to fully understand its broader effects. Much of the current knowledge is based on: Clinical and preclinical studies Controlled laboratory research Specific use-case investigations Further research is needed to expand understanding across different scenarios. Final Thoughts Tesamorelin is a synthetic peptide that works by stimulating natural growth hormone release. Its role in hormone signaling makes it an important subject in research focused on metabolism and cellular activity. At this stage, it remains part of ongoing scientific exploration, with continued studies helping to refine what we know about its function.
Exploring BPC-157 and Nitric Oxide: What Research Suggests

BPC-157 is a synthetic peptide that has been widely studied for its role in tissue repair and cellular signaling. One area that continues to draw attention is its potential interaction with nitric oxide, a molecule involved in blood flow and vascular function. Understanding this relationship helps researchers explore how peptides may influence key biological systems. What Is BPC-157? BPC-157 is a peptide derived from a protein found in gastric juice. In research settings, it is often studied for its connection to healing processes, including how tissues respond to stress or injury. Its stability and ability to interact with multiple pathways make it a frequent subject in peptide research. What Is Nitric Oxide? Nitric oxide is a signaling molecule produced naturally in the body. It plays a major role in regulating blood vessel function by helping vessels relax and expand. This process, known as vasodilation, allows for better blood flow and nutrient delivery throughout the body. How BPC-157 and Nitric Oxide May Be Connected Research suggests that BPC-157 may interact with pathways related to nitric oxide production and regulation. Some studies indicate that it may: Influence nitric oxide signaling pathways Support vascular response mechanisms Affect blood flow at the tissue level Interact with processes involved in repair and regeneration These interactions are of interest because nitric oxide is closely tied to how the body responds to injury and maintains circulation. Areas of Research Interest Researchers explore this connection across several areas: Vascular FunctionStudying how nitric oxide pathways affect blood flow and how peptides may influence those responses. Tissue Repair ProcessesUnderstanding how improved circulation and signaling contribute to healing at the cellular level. Cellular Signaling MechanismsAnalyzing how different pathways interact and regulate biological responses. What Current Research Suggests Most findings come from preclinical and laboratory studies. These suggest that BPC-157 may interact with nitric oxide systems in ways that influence vascular and cellular responses. However, results vary depending on the model and conditions used in research. Limitations and Ongoing Study Here’s the honest picture. While there is growing interest in the connection between BPC-157 and nitric oxide, large-scale human research is still limited. Most available data comes from: Animal studies Cell-based experiments Controlled laboratory research More research is needed to fully understand how these interactions behave across different contexts. Final Thoughts BPC-157 and nitric oxide are both important in understanding how the body regulates blood flow and tissue response. Their potential interaction makes them a valuable focus in ongoing peptide research. At this stage, this area remains under active investigation, with future studies expected to provide deeper insights into how these systems work together.
What Is Cagrilintide? A Research Overview

Cagrilintide is a synthetic peptide analog that has gained attention in research for its interaction with pathways related to appetite regulation and metabolic signaling. It is designed to mimic the activity of a naturally occurring hormone called amylin. Because of this, it’s often studied in connection with how the body manages energy balance and food intake. What Is Cagrilintide? Cagrilintide is an analog of amylin, a hormone that is normally released alongside insulin. Amylin plays a role in regulating appetite, slowing gastric emptying, and influencing how the body processes nutrients. By mimicking this hormone, Cagrilintide is studied to understand how these same pathways can be influenced in a controlled research setting. How It Works In research, Cagrilintide is examined for how it interacts with receptors involved in appetite and metabolic control. Some of its studied effects include: Influencing satiety signals Slowing the rate at which food leaves the stomach Affecting energy intake behavior Interacting with metabolic regulation pathways Rather than acting directly on energy production, it works through signaling mechanisms that help regulate intake and processing. Why It’s Being Studied Cagrilintide has become a focus in research because it targets specific pathways related to metabolic balance. Researchers are particularly interested in: Appetite RegulationUnderstanding how signals in the brain and body control hunger and fullness. Energy BalanceStudying how intake and expenditure are regulated through hormonal signaling. Peptide-Based PathwaysExploring how synthetic analogs can replicate and modify natural hormone activity. What Research Suggests So Far Most findings come from controlled studies and early-stage research. These suggest that Cagrilintide can influence appetite-related pathways through its interaction with amylin receptors. However, outcomes can vary depending on study design, dosage, and experimental conditions. Limitations and Current Understanding Here’s the honest take. While research is ongoing, there is still limited long-term data available across broader populations. Much of the current understanding is based on: Early-stage clinical research Laboratory studies Controlled experimental settings Further research is needed to fully understand its long-term behavior and broader implications. Final Thoughts Cagrilintide is a synthetic peptide designed to mimic amylin, making it a key subject in research focused on appetite and metabolic signaling. Its targeted mechanism has made it an area of growing interest. At this stage, it remains an active field of study, with ongoing research continuing to explore its full potential and applications.
What Is GHK-Cu? Understanding This Copper Peptide in Research
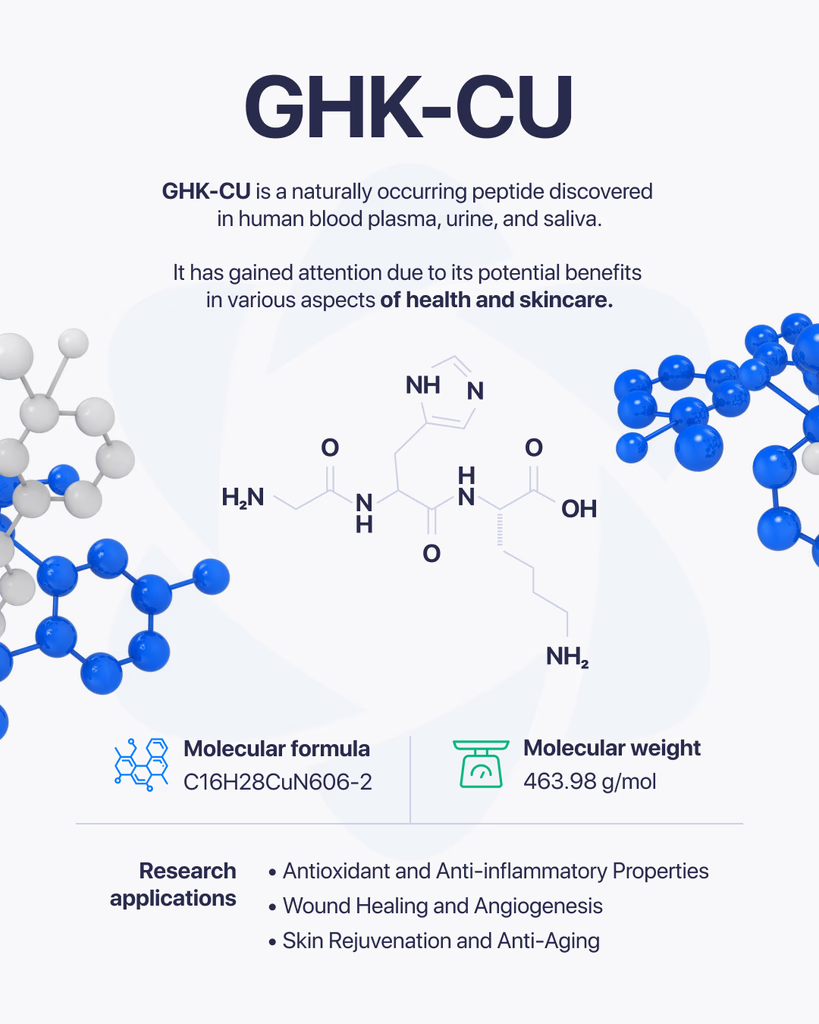
GHK-Cu is a naturally occurring peptide complex that has been widely studied for its role in cellular activity and tissue-related processes. It consists of a short peptide (GHK) bound to copper ions, which gives it unique properties in research settings. Because of this combination, it has become a point of interest in studies focused on cellular repair, signaling, and biological balance. What Is GHK-Cu Made Of? GHK-Cu is formed from a tripeptide made up of three amino acids: Glycine Histidine Lysine When this peptide binds with copper, it creates a complex that can interact with various biological systems. The copper component plays a key role in how the compound functions at the cellular level. How It Works in Research In research environments, GHK-Cu is studied for how it influences cellular communication and tissue-related processes. Some areas of focus include: Cellular repair mechanisms Regulation of gene expression Interaction with enzymes and proteins Support of structural components in tissues Its ability to bind copper allows it to participate in processes where metal ions are involved in biological activity. Why It’s Studied GHK-Cu has drawn attention because of its potential to influence multiple pathways at once. Researchers are interested in how it may support: Tissue-Related ProcessesStudying how cells repair and maintain structure. Cellular SignalingUnderstanding how signals are transmitted between cells. Structural Protein InteractionAnalyzing how it may affect components like collagen and other proteins. What Research Suggests Most available research comes from laboratory and preclinical studies. These suggest that GHK-Cu may play a role in regulating cellular processes and supporting tissue-related functions. However, outcomes can vary depending on conditions such as concentration, environment, and study design. Limitations and Current Understanding Here’s the honest picture. While GHK-Cu has been studied for many years, there is still ongoing research to fully understand all of its mechanisms and long-term effects. Most findings are based on: Cell-based studies Animal research Controlled laboratory environments More extensive research is needed to expand current knowledge. Final Thoughts GHK-Cu is a well-known peptide complex in research due to its connection to copper and its involvement in cellular processes. Its ability to interact with multiple biological pathways makes it an important subject of ongoing study. At this stage, it remains a key area of interest in peptide research, with continued investigation helping to uncover its full range of activity.
In Vitro vs In Vivo: What’s the Difference in Research?

If you’ve spent any time around scientific studies, you’ve probably come across the terms in vitro and in vivo. They sound technical, but the difference is actually straightforward once you break it down. These terms describe where and how research is conducted. What Does In Vitro Mean? In vitro refers to studies performed outside of a living organism. This usually means experiments carried out in a controlled environment like test tubes, petri dishes, or lab containers. The goal here is precision. Researchers isolate specific cells, tissues, or molecules to study how they behave under controlled conditions. Common examples include: Cell culture experiments Chemical interaction studies Early-stage compound testing This approach allows scientists to focus on one variable at a time without outside interference. What Does In Vivo Mean? In vivo refers to research conducted on a living organism. This could involve animal models or human studies, depending on the stage of research. Instead of isolating variables, in vivo studies observe how a compound behaves within a complete biological system. Examples include: Animal-based research models Clinical trials Whole-system biological studies This approach provides a more realistic picture of how something functions in the body. The Key Difference Here’s the simplest way to understand it: In vitro = outside the body, controlled lab environment In vivo = inside a living organism, real biological system One focuses on control. The other focuses on complexity. Why Both Are Important Neither method replaces the other. They serve different purposes and are often used together. Researchers typically start with in vitro studies to: Understand basic mechanisms Identify potential effects Test safety at a controlled level Once enough data is gathered, in vivo studies help: Confirm results in a full biological system Observe interactions across organs and systems Evaluate real-world biological responses Strengths and Limitations Each method has its advantages and trade-offs. In Vitro Strengths: High level of control Faster and more cost-effective Easier to isolate variables In Vitro Limitations: Lacks full biological complexity Results may not fully translate to living systems In Vivo Strengths: More realistic biological context Captures system-wide interactions Better for understanding overall effects In Vivo Limitations: More complex and time-consuming Harder to control every variable Final Thoughts In vitro and in vivo studies are both essential parts of the research process. One helps scientists understand the details in isolation, while the other shows how those details play out in a living system. Together, they provide a more complete picture, allowing researchers to move from controlled experiments to real biological insight.
Semax vs N-Acetyl Semax Amidate: What’s the Difference?

Semax and N-Acetyl Semax Amidate are closely related peptides often studied for their effects on the brain and cognitive processes. While they share a similar foundation, small structural differences between them can influence how they behave in research settings. Let’s break down what sets them apart. What Is Semax? Semax is a synthetic peptide derived from a fragment of adrenocorticotropic hormone (ACTH). It has been studied for how it interacts with brain signaling pathways, particularly those linked to cognitive function and neurotransmitter activity. In research, Semax is often explored for its potential influence on: Cognitive processes such as memory and focus Neurotransmitter regulation Brain-derived signaling pathways What Is N-Acetyl Semax Amidate? N-Acetyl Semax Amidate is a modified version of Semax. It includes two structural changes: An added acetyl group An amidated end These modifications are designed to improve stability and resistance to breakdown, allowing the peptide to remain active longer in research conditions. Key Differences Between the Two Here’s the simple comparison: Semax is the original peptide structure N-Acetyl Semax Amidate is a more stable, modified version Because of its modifications, N-Acetyl Semax Amidate is often studied for: Increased stability Longer duration in experimental conditions Potentially more consistent activity over time Why Stability Matters Peptides can degrade quickly depending on environmental conditions. By modifying the structure, researchers can observe how increased stability affects performance and interaction with biological systems. This is one of the main reasons analog versions like N-Acetyl Semax Amidate are developed and studied. Areas of Research Interest Both peptides are explored in similar research areas: Cognitive FunctionStudying how they may influence memory, learning, and focus-related pathways. Neurotransmitter ActivityAnalyzing interactions with systems such as dopamine and serotonin. Brain Signaling MechanismsUnderstanding how peptide-based compounds affect neural communication. What Research Suggests Most findings come from laboratory and early-stage studies. These indicate that both compounds interact with neurological pathways, but differences in stability may affect how long and how consistently they act. However, results can vary based on experimental conditions. Limitations and Ongoing Research Here’s the honest take. While both peptides are actively studied, there is limited large-scale clinical data comparing them directly. Most research is based on: Preclinical studies Controlled lab experiments Smaller-scale investigations More research is needed to fully understand how these differences translate across broader contexts. Final Thoughts Semax and N-Acetyl Semax Amidate are closely related peptides with similar research focus areas. The main difference lies in structural modifications that may influence stability and duration. For researchers, choosing between them often comes down to whether stability and extended activity are important for the specific study.
Investigating AOD-9604: A Review of Published Research
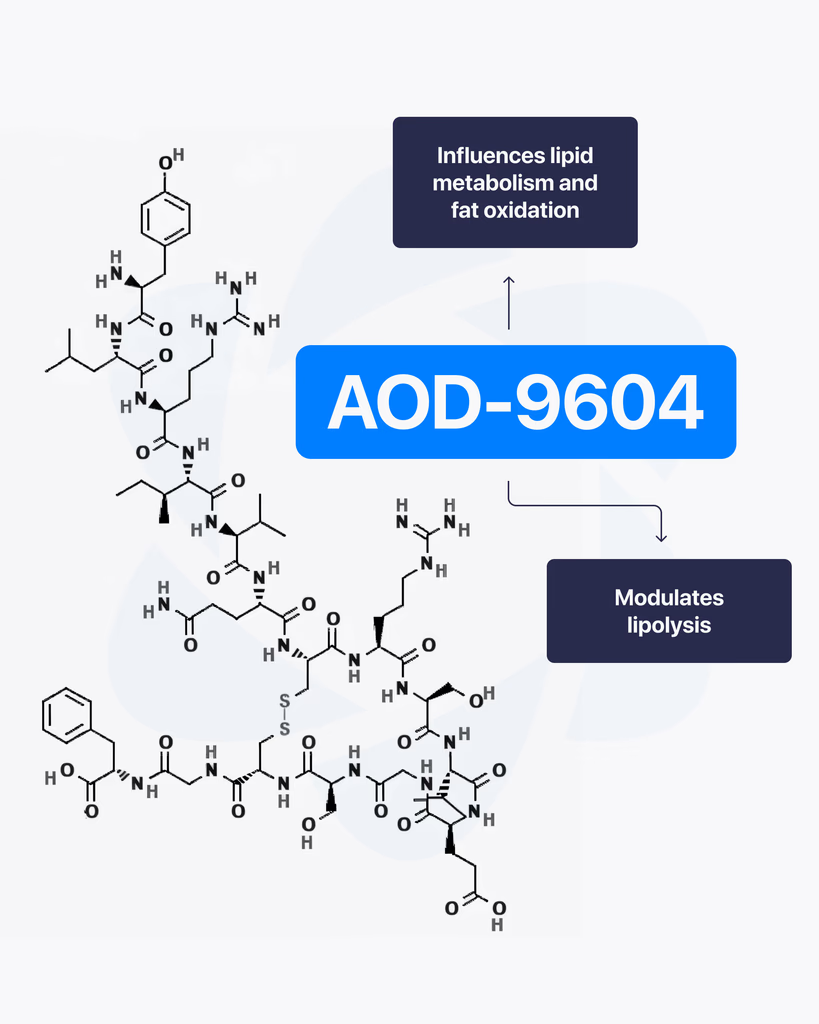
AOD-9604 is a synthetic peptide derived from a specific segment of growth hormone. Unlike full-length growth hormone, this fragment has been studied separately to understand how it interacts with metabolic pathways without triggering the broader effects associated with GH. Because of this targeted approach, it has become a subject of interest in peptide research focused on metabolism and cellular activity. What Is AOD-9604? AOD-9604 is a modified fragment of growth hormone, specifically designed to isolate certain biological actions. Researchers study it to examine how specific portions of larger hormones function on their own. Rather than acting as a full hormone, it is explored for its selective interaction with certain pathways. How It Works in Research In research settings, AOD-9604 is studied for how it may influence processes related to fat metabolism. Some studies suggest it interacts with pathways involved in: Lipolysis (breakdown of stored fat) Lipogenesis (formation of fat) Energy regulation at the cellular level The goal is to understand whether targeting specific mechanisms can produce focused effects without broader systemic impact. Areas of Research Interest Researchers have explored AOD-9604 across several areas: Metabolic ActivityStudying how it may influence energy use and fat-related pathways. Cellular-Level ProcessesAnalyzing how specific peptide fragments interact with metabolic signaling systems. Targeted Hormone PathwaysUnderstanding how isolating a segment of a larger hormone changes its behavior. What Published Studies Suggest Most available research includes laboratory studies and some early-stage clinical investigations. These suggest that AOD-9604 may interact with metabolic pathways in a more targeted way compared to full growth hormone. However, results vary depending on study design, dosage, and research conditions. Limitations and Current Understanding Here’s the honest picture. While AOD-9604 has been studied for its potential metabolic effects, the overall body of research is still developing. Much of the data comes from: Preclinical studies Controlled trials with limited scope Laboratory-based analysis More comprehensive research is needed to fully understand its long-term behavior and broader implications. Final Thoughts AOD-9604 represents an interesting direction in peptide research, where specific fragments of larger compounds are studied for targeted effects. Its connection to metabolic pathways continues to drive interest in ongoing studies. At this stage, it remains an active area of research, with further investigation needed to fully define its role and potential.
NAD+ Peptide Research: Mechanisms, Studies & Verification
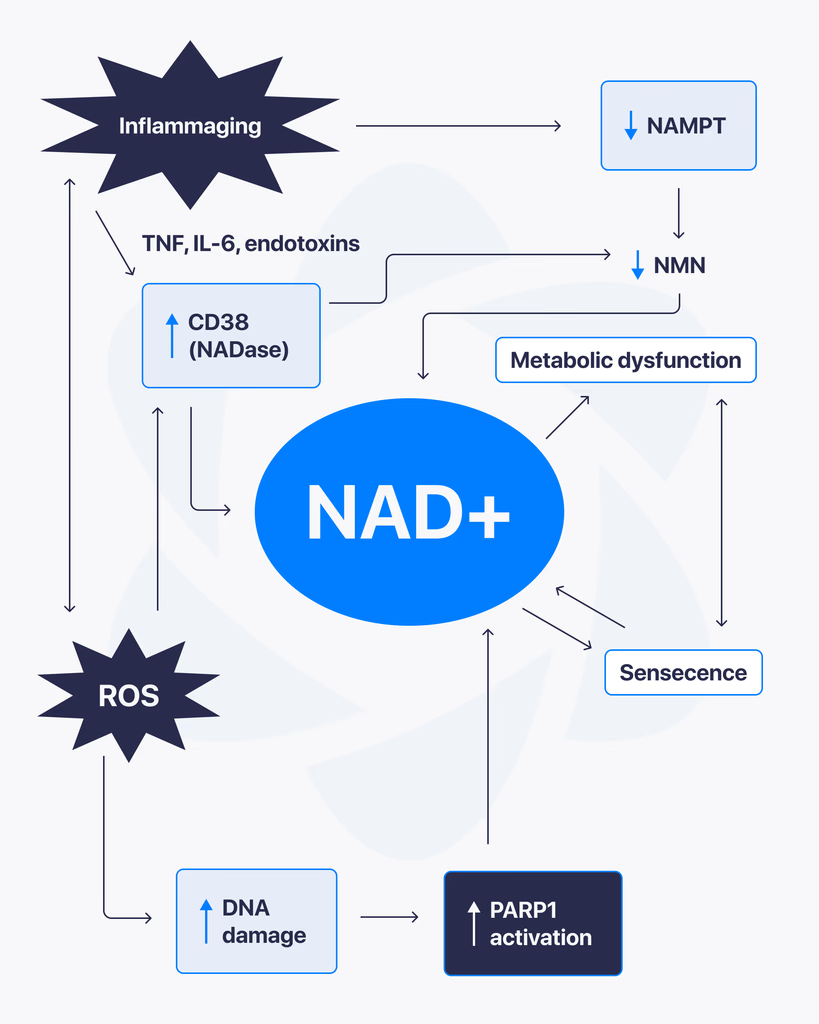
NAD+, short for nicotinamide adenine dinucleotide, is a coenzyme found in every living cell. It plays a central role in energy production and cellular function, which is why it has become a major focus in ongoing research. Scientists are studying how NAD+ levels influence different biological processes, especially those related to metabolism and cellular health. What Is NAD+? NAD+ is a molecule that helps transfer energy within cells. It’s involved in reactions that convert nutrients into usable cellular energy. As cells perform their functions, NAD+ cycles between two forms, helping drive essential processes that keep cells active and functioning. Why NAD+ Matters NAD+ is critical for several core cellular functions, including: Energy production within mitochondria DNA repair mechanisms Regulation of cellular stress responses Support of metabolic processes Because of its wide-ranging role, changes in NAD+ levels can affect how efficiently cells operate. NAD+ and Cellular Energy One of the main reasons NAD+ is studied is its connection to energy metabolism. It helps facilitate reactions that produce ATP, the primary energy source used by cells. Without sufficient NAD+, these processes become less efficient, which can impact overall cellular performance. What Research Is Exploring Researchers are examining how NAD+ interacts with different pathways inside the body. Some key areas of focus include: Mitochondrial FunctionUnderstanding how NAD+ supports energy production and efficiency within cells. Cellular Repair SystemsStudying its role in activating enzymes involved in DNA repair and maintenance. Metabolic RegulationExploring how NAD+ levels influence how the body processes and uses energy. What Studies Suggest So Far Most findings come from laboratory and preclinical research. These studies indicate that NAD+ plays a vital role in maintaining cellular function and responding to stress. There is also growing interest in how NAD+ levels change over time and how that may affect different biological systems. Limitations and Current Understanding Here’s the reality. While NAD+ is well understood at a biochemical level, many of its broader effects are still being researched. Most available data comes from: Cell-based studies Animal research Early-stage human investigations More comprehensive research is needed to fully understand long-term impacts and practical applications. Final Thoughts NAD+ is a key molecule in cellular energy and maintenance, making it an important subject in modern research. Its involvement in multiple biological pathways continues to drive interest across different scientific fields. At this stage, it remains an active area of study, with ongoing research working to better understand how it influences cellular function over time.
Acetic Acid 0.6% for Research Peptides: When to Use It, How It Works, and How OPTMZ Verifies the Solvent

Acetic acid is a widely used compound in chemical and laboratory settings, playing a role in everything from synthesis processes to analytical applications. As research continues to evolve, new methods are improving how acetic acid is produced, making the process more efficient, scalable, and environmentally conscious. What Is Acetic Acid? Acetic acid is a simple organic compound known for its role in various chemical reactions. In research and industrial settings, it’s commonly used as a solvent, reagent, and intermediate in synthesis. Its versatility makes it an important component in both small-scale laboratory work and large-scale production systems. Traditional Production Methods Historically, acetic acid has been produced through processes such as: Methanol carbonylation Oxidation of acetaldehyde Fermentation-based methods Among these, methanol carbonylation has become the dominant industrial approach due to its efficiency and scalability. What’s Changing in 2025 Recent developments are focused on improving sustainability, reducing waste, and increasing precision in production. Advanced CatalystsNew catalyst systems are being developed to improve reaction efficiency and reduce energy consumption. These catalysts help speed up production while minimizing unwanted byproducts. Greener Production TechniquesThere is a growing shift toward environmentally friendly methods, including bio-based production and reduced reliance on harsh chemicals. Process OptimizationModern systems are using improved monitoring and control technologies to ensure more consistent output and better resource management. Integration with Renewable InputsSome research is exploring the use of renewable feedstocks, aiming to reduce dependence on traditional petrochemical sources. Why These Innovations Matter Improving how acetic acid is produced has a direct impact on research and manufacturing. Better processes mean: Higher purity and consistency Reduced environmental impact Lower production costs over time More reliable supply chains For laboratories and industries that rely on precise chemical inputs, these improvements make a noticeable difference. Applications in Research Acetic acid continues to be used across various research areas, including: Chemical synthesis Analytical testing Peptide and organic compound preparation Buffer and solvent systems Its role as a foundational compound ensures it remains relevant as research techniques advance. Final Thoughts Acetic acid synthesis is evolving with a clear focus on efficiency and sustainability. Innovations in catalysts, production methods, and process control are shaping how this essential compound is produced and used. As research continues, these advancements are expected to further improve both quality and environmental impact, making acetic acid production more refined and future-ready.
How Does Sermorelin Work? GHRH Mechanism & Research Overview

By Dr. Leonard Haberman, Chief Science Officer, OPTMZ Peptides Published: April 2, 2026 · Last updated: April 16, 2026 Sermorelin is a 29-amino-acid synthetic peptide corresponding to residues 1–29 of growth hormone-releasing hormone (GHRH 1–44), the biologically active N-terminal fragment. In research models, sermorelin binds the GHRH receptor on anterior pituitary somatotrophs, activates adenylate cyclase, and triggers a cyclic AMP–protein kinase A (cAMP–PKA) signaling cascade that has been studied for its role in growth hormone gene transcription and release (Walker, 2006). Investigators have used sermorelin to examine GHRH-receptor pharmacology, downstream second-messenger dynamics, and — more recently — its interactions with cell proliferation pathways in oncology research models (Chang et al., 2021). This research overview summarizes what controlled studies have documented about sermorelin’s molecular mechanism, its half-life behavior in research contexts, what cell proliferation data has emerged, and how research-grade material is verified for laboratory use. What Is Sermorelin? Sermorelin is the truncated 1–29 amino acid sequence of native human GHRH, retaining the receptor-binding domain required for GHRH-R activation. The full-length endogenous peptide is 44 amino acids, but research has established that the first 29 residues are sufficient to reproduce the agonist activity of the parent molecule (Walker, 2006). The compound is supplied for research as a lyophilized powder requiring reconstitution in bacteriostatic water before use in cell-based or in-vivo research protocols. Structurally, sermorelin is classified as a GHRH analog rather than a growth hormone analog — a distinction that matters for experimental design. It does not interact directly with growth hormone receptors. Instead, it acts upstream at the pituitary level, making it a tool for studying GHRH-R signaling rather than peripheral GH-receptor activity. How Does Sermorelin Work? The GHRH Receptor Mechanism The mechanism of action observed in pre-clinical research follows a defined sequence at the molecular level: Receptor binding. Sermorelin binds to the GHRH receptor (GHRH-R), a class B G-protein-coupled receptor (GPCR) expressed on somatotroph cells of the anterior pituitary. G-protein coupling. Receptor activation triggers conformational changes that activate the Gαs subunit of the associated heterotrimeric G-protein. Adenylate cyclase activation. Gαs stimulates membrane-bound adenylate cyclase, which catalyzes the conversion of ATP to cyclic AMP (cAMP). PKA cascade. Elevated intracellular cAMP activates protein kinase A (PKA), which phosphorylates target proteins including the transcription factor CREB. Transcriptional regulation. Phosphorylated CREB binds the GH gene promoter, increasing transcription of growth hormone messenger RNA. Walker (2006) noted that this pathway “stimulates pituitary gene transcription of hGH messenger RNA, increasing pituitary reserve.” This signaling architecture is what makes sermorelin useful in research: it engages a well-characterized GPCR pathway with measurable second-messenger readouts (cAMP accumulation assays, CREB phosphorylation Western blots, GH ELISA from culture supernatant). A practical consequence observed in research contexts is that GH release driven by sermorelin remains under physiological feedback control by somatostatin and IGF-1, producing pulsatile rather than tonic patterns in animal models — a behavior distinct from exogenous recombinant GH administration (Walker, 2006). What Is the Half-Life of Sermorelin in Research Models? Sermorelin has a notably short plasma half-life — published research data places it at approximately 11–12 minutes in circulation, which is characteristic of GHRH-family peptides and reflects rapid enzymatic degradation by dipeptidyl peptidase-IV (DPP-IV) and other peptidases (Walker, 2006). For research workflows, this short half-life has several implications: Sampling windows are narrow. Time-course experiments measuring GH release require closely spaced sampling intervals (typically every 5–15 minutes for the first hour). Storage and reconstitution discipline matters. Once reconstituted, sermorelin should be maintained at 2–8°C and used within the verified stability window indicated on the batch certificate of analysis. Comparison studies use longer-acting analogs. Researchers comparing pulsatile versus sustained GHRH-R activation often pair sermorelin against modified GHRH analogs with extended half-lives (CJC-1295 and modified GRF 1–29 derivatives). What Has Research Shown About Sermorelin and Cell Proliferation? The most cited contemporary research on sermorelin and cellular proliferation is Chang et al. (2021), which used bioinformatic analysis (gene ontology and KEGG pathway enrichment) to examine sermorelin’s potential as a candidate for recurrent glioma research. The investigators reported that gene ontology analysis identified sermorelin as “closely related to cell proliferation functions” and proposed that the compound may inhibit tumor cell proliferation through cell cycle blocking mechanisms. Independent supporting work by Muñoz-Moreno et al. (2018) investigated GHRH-receptor antagonists in LNCaP and PC3 prostate cancer cell lines. While that work studied antagonists rather than agonists, it established that the GHRH-R signaling axis is functionally engaged in proliferative cell models — providing the receptor-pathway context that makes sermorelin’s effects in Chang’s bioinformatic analysis biologically plausible. It is important to characterize this body of work accurately: the cell proliferation data is bioinformatic and pre-clinical. There are no large-scale human studies investigating sermorelin’s effects on cellular proliferation. Researchers working in this area are typically running cell-line experiments, animal models, or computational analyses — not clinical trials. For laboratory teams designing experiments in this space, the key methodological considerations are: receptor expression validation in the chosen cell line (GHRH-R is not expressed at meaningful levels in all immortalized lines), batch-to-batch purity verification of the sermorelin used (impurities or degradation products can confound proliferation readouts), and inclusion of appropriate controls for cAMP-PKA pathway activation. How Does Sermorelin Compare to Other GHRH-Related Research Peptides? Within the GHRH-analog family, sermorelin is distinguished by its short half-life and its identity as the unmodified 1–29 sequence. Researchers comparing GHRH-pathway tools typically evaluate: CJC-1295 — a modified GRF 1–29 with a drug affinity complex (DAC) extension that significantly extends plasma half-life, used in research where sustained receptor engagement is the experimental variable. Tesamorelin — a stabilized GHRH 1–44 analog with an N-terminal trans-3-hexenoyl modification that resists DPP-IV cleavage. Ipamorelin — not a GHRH analog at all, but a ghrelin-receptor (GHS-R) agonist that converges on the same downstream output (GH release) through a parallel receptor system. This makes ipamorelin a useful comparator in studies dissecting the contributions of the two upstream pathways. The choice between these tools depends on the experimental question. Sermorelin’s short half-life and unmodified sequence make it the cleanest tool for studying acute,