By Dr. Leonard Haberman, Chief Science Officer, OPTMZ Peptides Published April 2, 2026 | Last updated April 16, 2026
Research Use Only. All products and information presented are intended for in vitro and pre-clinical research conducted by qualified professionals. Compounds discussed are not intended for human or animal consumption, clinical use, or therapeutic application. Statements have not been evaluated by the FDA. These materials are not intended to diagnose, treat, cure, or prevent any disease.
NAD+ (nicotinamide adenine dinucleotide) is a coenzyme — not technically a peptide, though commonly catalogued alongside research peptides in supplier catalogs — that functions as a central redox cofactor in cellular metabolism. Pre-clinical research has examined NAD+ and its precursors in cellular and animal models for roles in mitochondrial bioenergetics, sirtuin enzyme activity, PARP-mediated DNA repair, and aging biology. This article summarizes the mechanisms researchers have studied, the pre-clinical literature, and the verification standards that distinguish research-grade NAD+ batches.
What Is NAD+? (Definition, Classification, and Molecular Profile)
NAD+ is a dinucleotide composed of two nucleotides — nicotinamide and adenine — joined through their phosphate groups by a phosphoanhydride bond. It is present in every living cell and functions as an essential coenzyme in oxidation–reduction (redox) reactions central to energy metabolism (Cantó et al., 2015 PMID 26118927).
The molecular profile relevant to research applications:
Chemical name: Nicotinamide adenine dinucleotide
CAS Number: 53-84-9
Molecular formula: C₂₁H₂₇N₇O₁₄P₂
Molecular weight: 663.43 g/mol
PubChem CID: 5892
Classification: Pyridine nucleotide coenzyme
NAD+ exists in two interconverting forms — the oxidized form (NAD+) and the reduced form (NADH) — collectively referred to as the NAD+/NADH redox couple. The ratio between these two forms is one of the most studied indicators of cellular metabolic state in mitochondrial research (Cantó et al., 2015 PMID 26118927).
Is NAD+ Actually a Peptide?
No. NAD+ is a dinucleotide coenzyme, not a peptide. The two molecule classes are chemically distinct:
Peptides are short chains of amino acids linked by peptide (amide) bonds. Examples in research catalogs include BPC-157, TB-500, and Semaglutide.
NAD+ is composed of two nucleotides — nicotinamide mononucleotide and adenine mononucleotide — linked by a phosphoanhydride bond. It contains no amino acid residues and no peptide bonds.
The phrase “NAD+ peptide” persists in research-supplier catalogs and search behavior because NAD+ is frequently sold and discussed alongside research peptides, and because it shares some of the same buyer audience: researchers studying cellular signaling, metabolism, and aging biology. The classification matters for two practical research reasons:
Analytical methodology — purity verification of NAD+ uses HPLC methods optimized for nucleotides, which differ in column chemistry and mobile phase composition from typical peptide HPLC protocols.
Storage chemistry — NAD+ degradation pathways (hydrolysis of the phosphoanhydride bond, oxidation of the nicotinamide ring) differ from typical peptide degradation pathways (hydrolysis of peptide bonds, oxidation of methionine and cysteine residues). Research-grade NAD+ stability protocols reflect these differences.
For researchers ordering through supplier catalogs, the “NAD+ peptide” terminology is a search convention — the underlying compound is a coenzyme.
NAD+ vs NAD: What the “+” Denotes
The “+” in NAD+ indicates the oxidized form of the coenzyme, which carries a net positive charge on the nicotinamide ring’s nitrogen atom. The reduced form, NADH, carries no net charge after accepting two electrons and a proton during redox reactions.
In informal usage, “NAD” is sometimes written without specifying oxidation state. In the research literature, the oxidation state is almost always specified: NAD+ for the oxidized form, NADH for the reduced form, and the NAD+/NADH ratio for the dynamic equilibrium between them. The two forms cannot be substituted for one another in research applications — they are functionally distinct co-substrates for different enzyme classes.
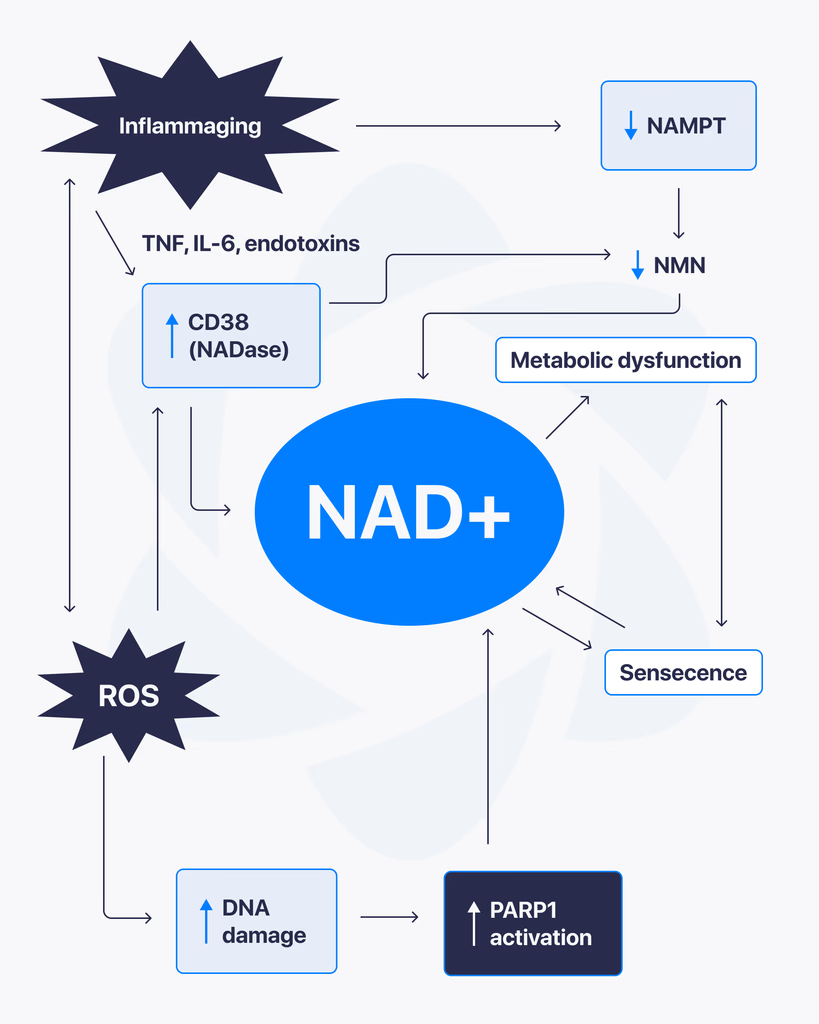
NAD+ Mechanisms Examined in Pre-Clinical Research
Pre-clinical research has examined NAD+ across four primary mechanistic categories. Each is supported by a substantial peer-reviewed literature.
Redox Cofactor Function in Mitochondrial Bioenergetics
NAD+ accepts electrons from substrates oxidized in glycolysis, the citric acid cycle, and fatty acid β-oxidation, becoming NADH in the process. NADH then donates electrons to Complex I of the mitochondrial electron transport chain, ultimately driving ATP synthesis. Cellular NAD+ availability is therefore a rate-limiting factor in mitochondrial energy production, and pre-clinical models have examined how perturbations to NAD+ pools affect oxidative phosphorylation efficiency (Cantó et al., 2015 PMID 26118927; Verdin, 2015 PMID 26785480).
Sirtuin Enzyme Co-Substrate Activity
Sirtuins (SIRT1–SIRT7) are a family of NAD+-dependent deacylases that have been extensively studied in cellular and animal models for their roles in metabolic regulation, transcriptional silencing, and stress responses. Each sirtuin-catalyzed deacylation reaction consumes one molecule of NAD+, producing nicotinamide and 2′-O-acyl-ADP-ribose. This stoichiometry makes sirtuin activity tightly coupled to cellular NAD+ availability (Imai & Guarente, 2014 PMID 24786309).
PARP Enzymes and DNA Repair Pathway Research
Poly(ADP-ribose) polymerases (PARPs) are NAD+-consuming enzymes activated by DNA strand breaks. Research has examined PARP1 in particular as a major NAD+ consumer in cellular models of genotoxic stress, where PARP hyperactivation can deplete cellular NAD+ pools (Rajman et al., 2018 PMC6342515).
NAD+ in Cellular Signaling
Beyond its classical role as a redox cofactor, NAD+ serves as a substrate for additional enzyme classes including CD38 (a cell-surface ectoenzyme studied in immunology and inflammation models) and SARM1 (studied in axonal degeneration models). These signaling roles are an active area of pre-clinical investigation (Verdin, 2015 PMID 26785480).
What NAD-Boosting Studies Have Examined in Animal Models
A substantial pre-clinical literature has examined whether elevating NAD+ levels — either through direct administration or through precursor supplementation — alters outcomes in animal models of metabolic dysfunction, mitochondrial impairment, and aging biology. The most comprehensive review of this literature to date is Rajman, Chwalek, and Sinclair (2018), which catalogued in vivo studies of NAD+ precursors including nicotinamide riboside (NR), nicotinamide mononucleotide (NMN), and direct NAD+ administration across rodent models (Rajman et al., 2018 PMC6342515).
The pre-clinical literature has examined:
Mitochondrial function in rodent models of muscle aging
Insulin sensitivity in diet-induced obesity models
Neuronal protection in models of axonal injury
Hepatic lipid metabolism in non-alcoholic fatty liver models
The translation of these pre-clinical findings to human contexts remains an open research question. Yoshino, Baur, and Imai (2018) provide a comprehensive review of NAD+ intermediate biology and the methodological challenges in moving from rodent studies to human investigations (Yoshino et al., 2018 PMID 29249689).
NAD+ vs Nicotinamide Riboside: Research Comparison
In supplier catalogs, NAD+ and nicotinamide riboside (NR) are frequently considered alongside one another. Mechanistically they are distinct:
NAD+ is the active coenzyme — directly consumed by sirtuins, PARPs, and electron transport chain dehydrogenases.
Nicotinamide riboside (NR) is a precursor that enters the salvage pathway, where nicotinamide riboside kinases convert it to nicotinamide mononucleotide (NMN), which is then adenylylated to NAD+.
Pre-clinical research has examined whether precursor supplementation (NR or NMN) is more effective at raising tissue NAD+ levels than direct NAD+ administration, with results that depend on tissue type, route of administration, and the model organism studied. Direct NAD+ administration is generally examined for in vitro applications and for cellular models, where membrane permeability and cellular uptake are controlled experimentally. Precursor supplementation has been more extensively studied in whole-animal models because precursors are more efficiently absorbed and distributed (Rajman et al., 2018 PMC6342515; Yoshino et al., 2018 PMID 29249689).
How Research-Grade NAD+ Is Verified for Purity and Identity
The defining characteristic of a research-grade NAD+ batch is documented, third-party analytical verification. At OPTMZ Peptides, every batch of NAD+ is tested by Krause Analytical — a DEA-registered, ISO/IEC 17025-certified laboratory in Austin, TX — using a defined seven-method analytical panel:
HPLC (high-performance liquid chromatography) for purity quantification — the primary method for detecting structurally similar impurities and degradation products such as ADP-ribose and nicotinamide.
Mass spectrometry (MS) for identity confirmation — verifies the molecular ion at m/z 663.43 and characteristic fragmentation pattern.
Endotoxin testing (LAL assay) — quantifies bacterial endotoxin contamination, relevant for cellular research applications where endotoxin can confound experimental results.
Heavy metals (ICP-MS) — quantifies elemental contaminants from synthesis or handling.
Microbial testing — detects bacterial and fungal contamination.
pH stability — confirms the lyophilized compound’s pH falls within specifications upon reconstitution.
Visual inspection — confirms physical appearance of the lyophilized powder.
OPTMZ’s minimum acceptance threshold is 98% HPLC purity. Batches below this threshold are rejected and never enter inventory. NAD+ batches have historically tested in the 98.5–99.9% range. The full Certificate of Analysis (COA) for every batch is published — searchable by batch number — in the OPTMZ Lab Analysis archive.
This is the structural difference between research-grade NAD+ from a verified-first supplier and NAD+ obtained without batch documentation. In research applications where compound purity directly affects experimental results — particularly in enzyme kinetics studies, cellular signaling assays, and any in vitro work where contaminants can confound readouts — published COA data is the only basis for rigorous batch-to-batch reproducibility.
For researchers who wish to verify NAD+ batches obtained from other sources, OPTMZ also operates an independent third-party peptide and compound testing service using the same Krause Analytical panel.
Research-Grade vs Pharmaceutical-Grade NAD+: Classification Differences
These are distinct regulatory and analytical classifications, and the difference matters for research procurement:
Research-grade (RUO — Research Use Only) materials are manufactured to defined analytical specifications (purity, identity, contamination thresholds) and are sold strictly for in vitro and pre-clinical research. They are not subject to FDA pharmaceutical manufacturing requirements (cGMP) and are not approved for human use.
Pharmaceutical-grade materials are manufactured under FDA cGMP requirements, typically for use in clinical drug formulations or as active pharmaceutical ingredients (APIs). They carry additional documentation, traceability, and process control requirements that research-grade materials do not.
OPTMZ Peptides supplies research-grade NAD+, manufactured and tested for in vitro and pre-clinical research use only. Per FDA Research Use Only classification (21 CFR 809.10(c)), research-grade materials carry the labeling restriction that they are “not for use in diagnostic procedures” and are intended exclusively for laboratory research. Researchers requiring pharmaceutical-grade material for clinical research applications should source from a cGMP-certified API manufacturer with the appropriate FDA documentation.
Storage and Handling Considerations for Research-Grade NAD+
NAD+ is supplied as a lyophilized powder. Standard research handling parameters relevant to NAD+ chemistry:
Lyophilized storage: -20°C, protected from light and moisture. Lyophilized NAD+ has demonstrated extended stability under these conditions in published stability literature.
Reconstituted storage: 2–8°C for short-term use; -20°C for longer-term storage of aliquots. Avoid repeated freeze–thaw cycles, which accelerate degradation.
Reconstitution solvent: Bacteriostatic water or sterile water, depending on the experimental application. The phosphoanhydride bond in NAD+ is susceptible to hydrolysis at extreme pH; neutral aqueous buffers are appropriate for most applications.
Stability monitoring: For long-term studies, periodic HPLC verification of working stocks is standard practice. Degradation products (ADP-ribose, nicotinamide) are detectable in the same HPLC method used for initial purity verification.
These are general research-handling parameters provided for laboratory context. Specific experimental protocols should follow the requirements of the cited literature for the application in question.
Dr. Leonard Haberman is Chief Science Officer at OPTMZ Peptides, overseeing analytical quality assurance and third-party laboratory partnerships with a focus on HPLC-based purity verification and research-grade peptide compound validation. All research peptides sold by OPTMZ Peptides are intended strictly for laboratory research use only.