By Dr. Leonard Haberman, Chief Science Officer, OPTMZ Peptides Published March 2, 2026 · Last Updated April 16, 2026
Research Use Only. The information in this article is provided for laboratory research and scientific education. The compounds discussed are not intended for human or animal consumption, clinical use, or therapeutic application. Statements on this site have not been reviewed by the FDA.
Ipamorelin is a synthetic pentapeptide studied in endocrine research for its selective stimulation of growth hormone release. Structurally defined as Aib-His-D-2-Nal-D-Phe-Lys-NH₂, it binds the growth hormone secretagogue receptor (GHS-R1a) — the same receptor activated by endogenous ghrelin — and has been characterized in published research as the first GHRP-class ligand with selectivity comparable to GHRH (Raun et al., 1998). This article reviews what the peer-reviewed literature has established about ipamorelin’s mechanism of action, its receptor selectivity, the key preclinical research areas it has been investigated in, and the analytical standards used to verify research-grade ipamorelin purity.
What Is Ipamorelin?
Ipamorelin is a synthetic five-amino-acid peptide (a pentapeptide) developed in the 1990s as part of research into growth hormone secretagogues — compounds that stimulate endogenous growth hormone (GH) release from the anterior pituitary. It was first characterized by Raun and colleagues at Novo Nordisk in 1998 and described as the first GHRP-receptor agonist whose selectivity for GH release was comparable to that of growth hormone-releasing hormone itself (Raun et al., 1998, PubMed).
In current peptide research contexts, ipamorelin is used as a pharmacological tool to investigate GH secretion, GHS-R1a receptor signaling, and related endocrine pathways. It is classified as a research compound and is not approved for clinical therapeutic use in any jurisdiction.
What Is the Molecular Structure of Ipamorelin?
Ipamorelin’s amino acid sequence is Aib-His-D-2-Nal-D-Phe-Lys-NH₂, where:
Aib is α-aminoisobutyric acid, a non-proteinogenic amino acid that introduces conformational rigidity
His is L-histidine
D-2-Nal is D-2-naphthylalanine, a non-natural D-amino acid
D-Phe is D-phenylalanine
Lys-NH₂ is C-terminal lysine amide
The inclusion of D-amino acids and the unnatural Aib residue is deliberate. These substitutions increase resistance to enzymatic degradation and alter the peptide’s secondary structure, producing a compact, receptor-selective ligand. The molecular formula is C₃₈H₄₉N₉O₅ with a molecular weight of approximately 711.85 g/mol.
Structurally, ipamorelin is derived from the GHRP-1 lineage but has been optimized to reduce off-target receptor engagement. This optimization is the basis of the selectivity profile described below.
How Does Ipamorelin Stimulate Growth Hormone Release?
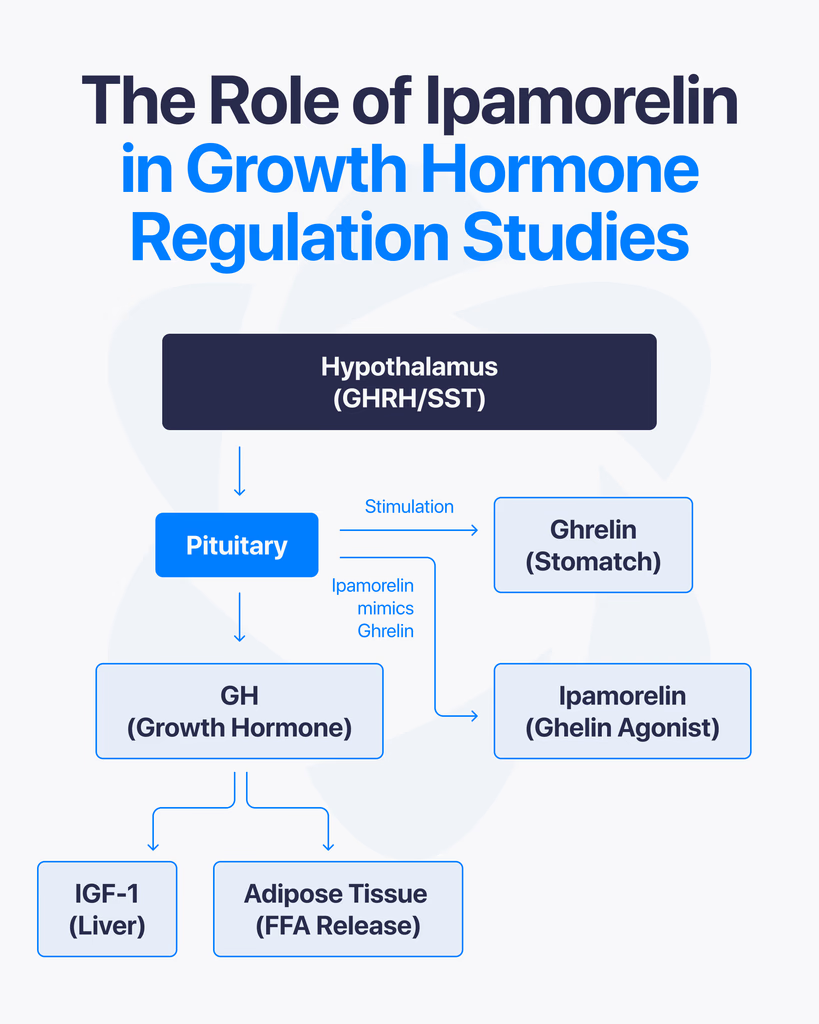
Ipamorelin is an agonist of the growth hormone secretagogue receptor type 1a (GHS-R1a) — a G-protein-coupled receptor (GPCR) expressed on somatotroph cells of the anterior pituitary and in regions of the hypothalamus. The GHS-R1a is the same receptor that binds endogenous ghrelin.
Upon binding, ipamorelin activates the Gq/11 pathway, triggering phospholipase C (PLC) activity, inositol triphosphate (IP₃) generation, and intracellular calcium mobilization. This cascade results in the release of stored growth hormone from somatotroph secretory vesicles into systemic circulation. In pharmacokinetic-pharmacodynamic modeling performed in healthy human volunteers, ipamorelin administration produced a rapid, dose-dependent increase in serum GH, with measurable GH elevation occurring within minutes of subcutaneous or intravenous delivery (Gobburu et al., 1999, PubMed).
Unlike synthetic growth hormone itself, which directly supplements circulating GH levels, ipamorelin stimulates the pituitary’s own GH-releasing machinery. Research on GHS-R1a agonists has shown that this mode of action preserves the pulsatile pattern of GH secretion — a pattern considered physiologically important in native endocrine regulation.
What Makes Ipamorelin Selective Compared to Other GHRPs?
The defining characteristic of ipamorelin in the peer-reviewed literature is its selectivity — specifically, its stimulation of GH release without significant concurrent elevation of adrenocorticotropic hormone (ACTH), cortisol, or prolactin.
Earlier growth hormone secretagogues, including GHRP-6 and GHRP-2, reliably stimulate GH release but also produce measurable increases in ACTH and cortisol, and in some studies prolactin as well. This cross-reactivity has historically complicated the use of these earlier peptides as clean research tools for isolating GH-specific effects.
Raun et al. (1998) demonstrated in both in vitro and in vivo models that ipamorelin, administered at doses that produced GH responses equivalent to GHRP-6, did not significantly elevate ACTH or cortisol. Johansen et al. (1999) extended these observations and characterized ipamorelin’s receptor binding kinetics, confirming that the selectivity appears to originate from the peptide’s specific binding profile at GHS-R1a and its lack of engagement with receptors responsible for corticotropin release (Johansen et al., 1999, PubMed).
For researchers, this matters because it allows the study of GH-dependent pathways with fewer confounding endocrine variables. In bone formation research, for example, Svensson et al. (2000) were able to examine ipamorelin’s influence on bone mineral content in adult rats without simultaneously introducing the catabolic effects of elevated glucocorticoids that would have been expected with less-selective secretagogues (Svensson et al., 2000, PubMed).
What Did the Original Discovery Research Establish?
Two foundational publications anchor the scientific understanding of ipamorelin.
Raun K, Hansen BS, Johansen NL, et al. (1998). Ipamorelin, the first selective growth hormone secretagogue. European Journal of Endocrinology, 139(5):552-561. (PubMed 9849822) — the foundational discovery paper. It established ipamorelin’s potency and efficacy in vitro (using isolated pituitary cells) and in vivo (in swine and rats), benchmarked it against GHRP-6, and documented the distinctive selectivity profile. The paper has been cited more than 180 times in subsequent literature.
Johansen PB, Segev Y, Landau D, et al. (1999). Ipamorelin, a new growth-hormone-releasing peptide: Pharmacokinetic and pharmacodynamic properties. (PubMed 10373343) — extended the receptor characterization, established pharmacokinetic parameters in rats, and confirmed in additional models that GH release was not accompanied by ACTH or prolactin elevation.
Subsequent research has built on these foundations. Gobburu and colleagues (1999) developed a pharmacokinetic-pharmacodynamic model based on data from human volunteers that described ipamorelin’s GH response curve across a range of doses. The 1998 and 1999 papers remain the primary citations in virtually all contemporary ipamorelin research.
How Does Ipamorelin Interact with Somatostatin and Endocrine Feedback?
Native GH secretion is regulated by a two-signal system: growth hormone-releasing hormone (GHRH) from the hypothalamus stimulates release, and somatostatin (also hypothalamic) inhibits it. Endogenous ghrelin — and, by extension, GHS-R1a agonists like ipamorelin — acts on a third, parallel pathway.
Research has investigated how ipamorelin interacts with this feedback system. GHS-R1a activation appears to both stimulate somatotroph GH release directly and attenuate the inhibitory influence of somatostatin on pituitary output. This dual action is one proposed explanation for why GHS-R1a agonists can produce sharp, pulsatile GH responses rather than the sustained, plateau-like elevation produced by direct GH administration.
Because ipamorelin works through this endogenous signaling axis, negative feedback mechanisms — elevated GH triggering increased somatostatin tone and suppressing further release — remain intact. This is of particular methodological interest for researchers modeling native endocrine physiology, as opposed to simple GH replacement.
What Research Applications Has Ipamorelin Been Investigated In?
Preclinical research using ipamorelin has examined several distinct pathways. The following are research contexts that have appeared in the peer-reviewed literature:
Bone mineral content in animal models. Svensson et al. (2000) examined the effects of chronic ipamorelin administration on bone mineral content in adult rats, observing increases relative to controls and providing a research framework for studying GH–IGF-1 axis activity on skeletal tissue independent of glucocorticoid confounds (PubMed 10828840).
Gastric motility and postoperative ileus models. Greenwood-Van Meerveld et al. (2012) characterized ipamorelin’s prokinetic activity in rodent models of postoperative ileus, where the peptide accelerated gastric emptying through GHS-R1a activation of cholinergic pathways in the enteric nervous system (PubMed 27186127).
Pancreatic insulin secretion. Adeghate and Ponery (2004) investigated ipamorelin’s effect on insulin release from isolated pancreatic tissue in normal and diabetic rat models, reporting calcium- and adrenergic-dependent increases in insulin secretion (Neuroendocrinology Letters, 25(6):403-406).
GH pulsatility research. Multiple studies have used ipamorelin as a pharmacological probe to examine the temporal dynamics of GH release, given that its selectivity makes it suitable for isolating GH-axis variables without confounding hormonal noise.
These are pre-clinical research contexts. Ipamorelin has not been approved for clinical therapeutic use. Some early-phase human trials (including one in postoperative ileus) did not meet primary clinical endpoints, though underlying pharmacological signals remained measurable. The compound’s current utility is as a research ligand, not as a therapeutic agent.
How Is Research-Grade Ipamorelin Verified for Purity?
For research applications, the quality of the peptide material used directly determines the validity of experimental results. Impurities — whether truncated sequences from incomplete solid-phase synthesis, racemized amino acids, residual solvents, or endotoxins — can produce confounded data and irreproducible results. This is why analytical verification matters.
The OPTMZ Peptides testing panel, performed for every batch by Krause Analytical — a DEA-registered, ISO/IEC 17025-certified laboratory in Austin, TX — includes the following methods for ipamorelin and all catalog compounds:
High-Performance Liquid Chromatography (HPLC) — quantifies purity by separating the target peptide from truncated or deletion sequences, typically using a C18 reverse-phase column and UV detection at 214 nm.
Mass Spectrometry (MS) — confirms molecular identity by matching the measured mass to the theoretical mass of the target sequence.
Endotoxin testing (LAL) — measures bacterial endotoxin contamination, important for any peptide that will be used in cell culture research.
Heavy metals screening (ICP-MS) — detects trace heavy metal contamination.
Microbial testing — confirms sterility of the lyophilized powder.
pH stability testing — verifies reconstitution behavior.
Visual inspection — confirms appearance and integrity of the lyophilized product.
OPTMZ’s minimum acceptance threshold is 98% HPLC purity. Batches testing below this threshold are rejected and never enter inventory; most batches test in the 98.5–99.9% range. The Certificate of Analysis for every production batch is published at the OPTMZ Lab Analysis vault and is linked directly from each product page. For detailed methodology, see the OPTMZ testing methodology page.
Research-grade Ipamorelin from OPTMZ Peptides ships as a lyophilized powder and is typically reconstituted with bacteriostatic water for laboratory preparation. The CJC-1295 + Ipamorelin research blend is also available for investigators studying combined GHRH-analog and GHS-R1a agonist pharmacology.
Dr. Leonard Haberman is Chief Science Officer at OPTMZ Peptides, overseeing analytical quality assurance and third-party laboratory partnerships with a focus on HPLC-based purity verification and research-grade peptide compound validation. All research peptides sold by OPTMZ Peptides are intended strictly for laboratory research use only.