By Dr. Leonard Haberman, Chief Science Officer, OPTMZ Peptides Published: March 2, 2026 | Last Updated: April 16, 2026
Peptide synthesis is the controlled laboratory process by which researchers build peptide chains from individual amino acid subunits. Rather than isolating peptides from biological tissues, synthetic production allows precise sequence specification — making it the standard method for producing research-grade compounds used in pre-clinical and in vitro studies.
Understanding how research peptides are synthesized helps researchers make better decisions about supplier selection, purity interpretation, and storage protocols. This article covers the core synthesis method, the purification steps that follow, and the analytical testing that determines whether a finished batch meets research-grade standards.
What Is Peptide Synthesis?
Peptide synthesis is the process of forming peptide bonds between amino acids in a defined sequence. In biological systems, this occurs through ribosomal translation of mRNA sequences. In laboratory settings, synthesis is performed chemically — allowing researchers to specify any sequence, introduce non-natural amino acids, or produce compounds that do not exist in sufficient quantities in natural sources.
The peptide bond itself is a covalent amide linkage between the carboxyl group of one amino acid and the amino group of the next. Each addition extends the chain by one residue. A ten-residue peptide requires nine successful bond formations, each carried out under controlled conditions (Merrifield, 1963 — PubMed).
The precision of the final sequence determines the compound’s research relevance. A single incorrect residue, deleted residue, or epimeric impurity can alter receptor binding, biological stability, or assay behavior. This is why synthesis methodology and post-synthesis verification are inseparable from research utility.
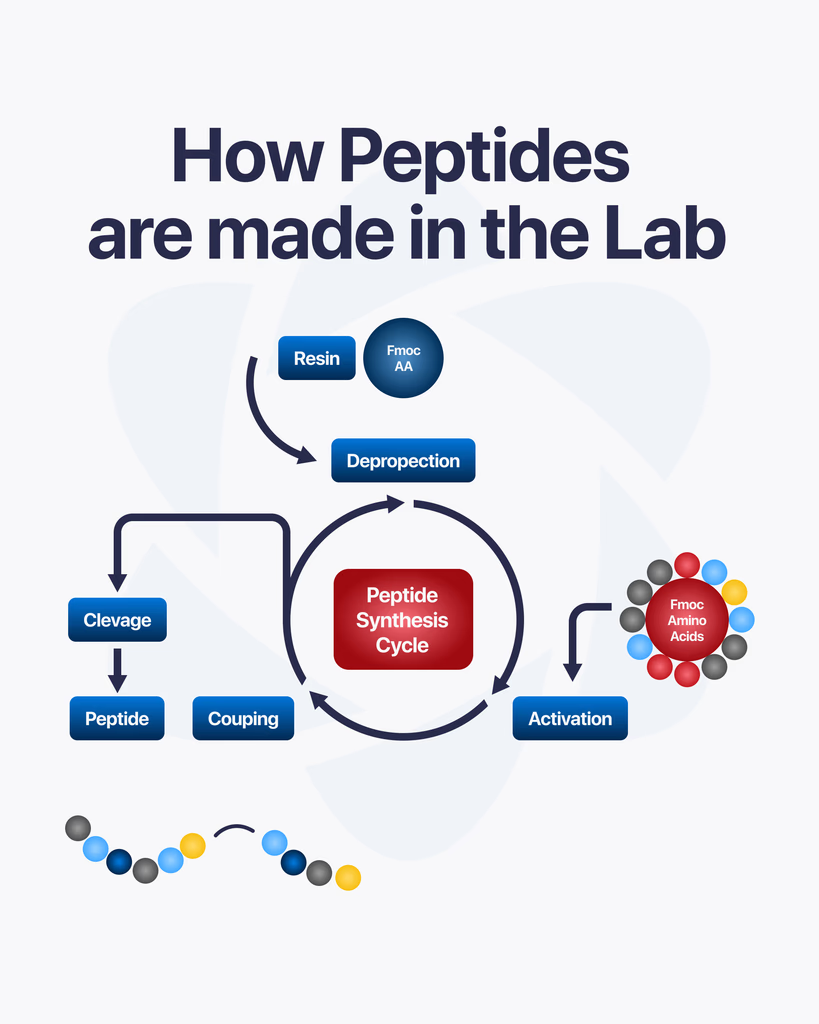
What Is Solid-Phase Peptide Synthesis (SPPS)?
Solid-phase peptide synthesis (SPPS) is the dominant method for producing research-grade peptides. Introduced by Robert Bruce Merrifield in 1963, the technique anchors the growing peptide chain to an insoluble polymer resin, which allows excess reagents and byproducts to be washed away between each coupling step (Merrifield, 1963 — PubMed).
How Does SPPS Work?
The synthesis proceeds through a repeating cycle:
1. Resin loading. The C-terminal amino acid of the target sequence is attached to the solid resin support via a linker. The resin physically anchors the chain throughout synthesis, enabling filtration-based purification between steps.
2. Deprotection. Amino acids carry protecting groups on their reactive side chains and N-terminus. Before each coupling step, the N-terminal protecting group is removed — a process called deprotection — exposing the free amino group for the next reaction.
3. Coupling. The next amino acid in the sequence is added in excess with a coupling reagent. The coupling reagent activates the incoming amino acid’s carboxyl group, enabling it to react with the deprotected N-terminus on the resin-bound chain. This forms a new peptide bond.
4. Washing. Excess reagents, activating agents, and byproducts are washed away with solvent. The resin retains the growing peptide chain.
5. Iteration. The deprotection → coupling → washing cycle repeats for each amino acid in the sequence, proceeding from C-terminus to N-terminus.
6. Cleavage and global deprotection. When the full sequence has been assembled, the completed peptide is cleaved from the resin. Simultaneously, remaining side-chain protecting groups are removed, yielding the free peptide in solution.
The primary advantage of SPPS over solution-phase synthesis is the ability to use large excesses of coupling reagents to drive reactions to completion — a requirement when even a small percentage of incomplete couplings results in deletion sequences or truncated impurities that reduce the final purity.
Two main SPPS strategies exist based on the N-terminal protecting group chemistry used: Boc (t-Boc) SPPS and Fmoc SPPS. Fmoc chemistry has become the standard for most research peptide synthesis due to its milder deprotection conditions and compatibility with a wider range of side-chain protecting groups (Carpino and Han, 1972 — PubMed).
Why Do Incomplete Coupling Reactions Matter for Research?
Each coupling step in SPPS does not achieve 100% efficiency. A typical coupling reaction yields 98–99.9% efficiency depending on the amino acid, the sequence context, and the coupling reagent used. Across a long sequence, these small inefficiencies accumulate.
For a 20-residue peptide synthesized at 99% per-step coupling efficiency, the theoretical yield of the fully correct sequence is approximately 82% (0.99²⁰). The remaining fraction consists of deletion peptides — sequences where one or more amino acids have been skipped — and truncated sequences that terminated early.
These impurities do not always share the same biological behavior as the target peptide. In research applications where receptor selectivity, binding affinity, or downstream signaling effects are being studied, impurity profiles that deviate significantly from the intended sequence can introduce confounding variables into experimental results (Chan and White, 2000 — PubMed).
This is why purification is not optional for research-grade compounds.
How Is a Research Peptide Purified After Synthesis?
The crude peptide cleaved from the resin contains the target compound alongside synthesis byproducts, incomplete sequences, and protecting group fragments. Purification removes these impurities to yield a product that meets defined purity thresholds for research use.
Reversed-Phase High-Performance Liquid Chromatography (RP-HPLC)
Reversed-phase HPLC (RP-HPLC) is the standard purification method for research peptides. The crude peptide mixture is dissolved and injected onto an HPLC column packed with a hydrophobic stationary phase (typically C18). As a mobile phase gradient of water and organic solvent (usually acetonitrile) moves through the column, peptide components separate based on their hydrophobicity.
The target peptide elutes at a characteristic retention time, and the corresponding fraction is collected. Other fractions — containing deletion sequences, oxidation products, and protecting group adducts — are discarded.
Preparative RP-HPLC can achieve purity levels exceeding 98% for most research peptide sequences. The resulting material is lyophilized (freeze-dried) to produce a stable white to off-white powder suitable for storage and research use.
How Is Peptide Purity Analytically Verified?
Purification produces a compound of defined purity. Analytical testing confirms that purity and verifies the compound’s identity. For research-grade peptides, this is not a quality assurance formality — it is the primary evidence that the compound in the vial is what the label states.
What Does HPLC Purity Testing Measure?
Analytical HPLC (distinct from preparative HPLC used for purification) quantifies the proportion of the total UV-absorbing material at 214–220 nm that corresponds to the target peptide peak. The result is expressed as a percentage of peak area.
A purity of 98% by HPLC means that 98% of the UV-absorbing material eluting from the column is the target peptide, with 2% comprising all other species — including synthesis impurities, degradation products, and residual protecting groups.
Research-grade peptides are generally specified at ≥95% HPLC purity. Suppliers setting a minimum threshold of 98% are applying a more stringent standard. OPTMZ Peptides applies a ≥98% minimum, with most batches testing between 98.5–99.9% by HPLC analysis performed by Krause Analytical (DEA-registered, ISO/IEC 17025-certified, Austin, TX). Batches below 98% are rejected and do not enter inventory.
What Does Mass Spectrometry Confirm?
Mass spectrometry (typically electrospray ionization, ESI-MS) confirms the molecular identity of the peptide by measuring its molecular weight. The observed mass-to-charge ratio (m/z) is compared against the theoretical molecular weight of the target sequence.
A correct mass confirmation verifies that the compound has the intended molecular formula — ruling out truncated sequences of similar length, oxidation artifacts that would shift the mass by +16 Da, or sequence scrambling that would produce a different molecular weight despite similar chromatographic behavior.
Identity confirmation by mass spectrometry is a required test in addition to HPLC purity — the two together constitute the minimum standard for research-grade compound verification. Batch-level identity confirmation data for every OPTMZ Peptides compound is published in the COA Vault.
What Additional Tests Are Applied to Research Peptides?
Beyond HPLC purity and mass spectrometry identity, research-grade peptides intended for cell-based or in vivo model research applications are evaluated for contaminants that can interfere with biological assays:
Endotoxin testing (LAL method): Detects bacterial endotoxins (lipopolysaccharides) introduced during synthesis or handling. Endotoxin contamination can activate innate immune pathways in cell culture models, confounding any experiment that measures inflammatory signaling.
Heavy metals (ICP-MS): Identifies trace metal contamination from synthesis reagents or equipment.
Microbial testing: Confirms absence of viable microbial contamination.
pH and visual inspection: Confirms solution stability and appearance of the reconstituted peptide.
Each of these tests is performed by Krause Analytical on every batch before release. The full test panel for any compound can be accessed via the OPTMZ COA Vault by searching the batch number printed on the vial label.
What Does a Certificate of Analysis (COA) Document?
A Certificate of Analysis (COA) is the primary documentation artifact for a research peptide batch. It records the testing performed, the methods used, the results obtained, and the laboratory that performed the testing.
A COA issued by an independent, third-party laboratory carries significantly more evidentiary weight than a COA prepared by the vendor internally. Key items that should appear on a research-grade COA include:
Peptide name and sequence
Batch or lot number
Manufacturing date and test date
HPLC purity result with chromatogram
Mass spectrometry result with spectrum
Endotoxin result (if tested)
Heavy metals result (if tested)
Identity of the testing laboratory, including any accreditation (ISO/IEC 17025)
The ability to cross-reference a vial’s batch number against a published COA archive — and verify that the data was produced by an accredited third-party lab — is the operational definition of supplier transparency in research peptide procurement. A detailed guide to reading peptide COA documentation is available at How We Test.
From Synthesis to Research: Why Supplier Verification Matters
The synthesis process described above produces a peptide. Purification increases the purity of that peptide. Analytical testing documents its actual composition. Only the third step creates verifiable evidence — and only when that testing is performed independently and the results are published.
A researcher selecting a peptide supplier should be able to answer three questions about any compound they intend to use:
What purity was this specific batch tested at, and by whom?
Was identity confirmed by mass spectrometry for this batch?
Can I access the original COA, not a summary created by the vendor?
If any of these questions cannot be answered with a direct link to primary data, the synthesis and purification process — however rigorous — remains unverified for the specific batch in hand. Synthesis methodology describes a process. Batch-specific COA data provides evidence that the process was executed correctly for the compound being ordered.
All OPTMZ Peptides batch COA documents are publicly accessible at the COA Vault, searchable by batch number printed on each vial label.
Dr. Leonard Haberman is Chief Science Officer at OPTMZ Peptides, overseeing analytical quality assurance and third-party laboratory partnerships with a focus on HPLC-based purity verification and research-grade peptide compound validation. All research peptides sold by OPTMZ Peptides are intended strictly for laboratory research use only.